Knowde Enhanced TDS
Identification & Functionality
- Active Component
- Chemical Family
- Ingredient Name
- Ingredient Origin
- Animal Feed & Nutrition Functions
- Cosmetic Ingredients Functions
- Food Ingredients Functions
- Pharma & Nutraceuticals Functions
- Ingredients
- Siberian Larch Dry Extract
- Technologies
- Product Families
- Chemical Composition
Flavonoids - dihydroquercetin (90-98%), dihydrokaempferol, kaempferol, naringenin, saponins.
Target Component:
Taxifolin (dihydroquercetin) - non less than 90%;
Taxifolin (dihydroquercetin) - non less than 92%:
Taxifolin (dihydroquercetin) - non less than 94%;
Taxifolin (dihydroquercetin) - non less than 96%;
Taxifolin (dihydroquercetin) - non less than 98%.
Features & Benefits
- Benefit Claims (Health)
- Labeling Claims
- Food Ingredients Features
- Product Background
Siberian larch is highly valued for its medicinal properties, is a source of invaluable components for such fields of activity as medicine, veterinary medicine, cosmetology, food industry. One of these components is taxifolin (dihydroquercetin), a powerful natural antioxidant flavonoid obtained by extraction from the butt of Siberian larch
- General Effects
- The antioxidant effect of dihydroquercetin is associated with its ability to accept free radicals and / or chelate metal ions that catalyze oxidation processes.
- Anti-inflammatory activity is due to the antioxidant effect and inhibitory effect on enzymes (enzymes) produced during inflammation, subsequent inhibition of the production of inflammatory mediators, regulating and modulating action on key functional systems of cells, organs and tissues of the body.
- Has a capillary-protective effect, reduces the permeability and fragility of capillaries, improves microcirculation, helps to inhibit the action of the enzymes hyaluronidase and collagenase, which loosen the connective tissue of the walls of blood vessels and other systems, but activates proline hydroxylase, which contributes to the "maturation of collagen, thus elasticity and normalization of vascular permeability - Helps to improve the rheological properties of blood, central and peripheral hemodynamics.
- Blocks the formation of complexes of acetaldehyde, which is a product of alcohol metabolism, with amino acids and albumin protein, thereby reducing the toxic effect of alcohol on human tissues and organs.
- Promotes an increase in exercise tolerance, provides an increase in the rehabilitation effect after prolonged physical exertion in athletes.
- Product Features
Botanical Origin
Larix sibirica Ledeb.
Plant Part
Wood (butt)
Mesh
200
Appearance
Fine powder from white to pale yellow color with a characteristic taste and smell.
Shelf Life
3 years
Packaging
Ingredients are packed into double bags to avoid moisture condensation. During shipment bags are stored into double-double carton boxes.
Weight
10 kg
Manufacture
We have built a full production cycle - from the search and collection of raw materials, the manufacture of the product to the delivery of the order to the client's territory. This
guarantees you stable prices, stable quality and continuous deliveries. We focus on the needs of our customers and, upon request, carry out additional analyzes, develop
the necessary documentation, and we produce extracts and powders with the characteristics you need.Verification
We check raw materials and final products with the help of laboratory tests at every stage from raw material preparation to finished extracts and powders. The supplied
products comply with the EU safety and quality standards.Safety & Quality
- GMO Free
- BSE/TSE free
- Allergen free
Applications & Uses
- Applications
- Food & Nutrition Applications
- Applications
Industry
- Cosmetic
- Pharmaceutical industry
- Food supplements
- Food and drinks
- Feeds
- Healthy nutrition
Indications
- In diseases of the cardiovascular system: as part of the complex therapy of ischemic heart disease (unstable angina pectoris), atrial arrhythmia, atherosclerosis, hypertension, in violation of capillary permeability and peripheral circulation
- In diseases of the gastrointestinal tract: gastric ulcer and duodenal ulcer, prolonged intestinal dysfunctions of unknown etiology, erosive and atrophic gastritis.
- In diseases of the respiratory system: bronchopulmonary diseases, including pneumonia, chronic obstructive bronchitis, bronchial asthma (Infectious form) in the acute stage.
- Type II diabetes mellitus,
- For the prevention of oncological diseases, for the rehabilitation of the body after illnesses, for the prevention and elimination of the consequences of alcohol poisoning, nicotine, low-quality food products, other organic and inorganic poisons
Contrindictions
Individual Intolerance
Properties
- Flavor
- Characteristic
- Typical Properties
- Physico-Chemical Properties
- Microbiological Values
- Allergen Declaration EU
Allergens Direct Incorporation Cross Contamination Presence on the production line Presence on the production workshop Presence on the production factory Cereals containing gluten (1) and
products thereofNO NO NO NO Crustaceans and products
there ofNO NO NO NO Eggs and products thereof NO NO NO NO Fish and products thereof (2) NO NO NO NO Peanuts and products there of NO NO NO NO Soybeans and products there of
(3)NO NO NO NO Milk and products thereof
(including lactose) (4)NO NO NO NO Nuts (5) or products thereof NO NO NO NO Celery and products there of NO NO NO NO Mustard and products there of NO NO NO NO Sesame seeds and products
there ofNO NO NO NO Sulfur dioxide and sulphites at
concentrations of more than 10
mg/kg or 10 mg/l expressed as
SO2NO NO NO NO Lupines and products thereof NO NO NO NO Mollusk and product thereof NO NO NO NO (1) Cereals which contain gluten (i.e.w heat, rye, barley, oats, spelt, kamut or their hybridized strains) except: w heat-based glucose syrups including dextrose, wheat-based maltodextrins, glucose syrups based on barley, cereals used for making distillates or ethyl alcohol of agricultural origin for spirit drinks and other alcoholic beverages.
(2) Except: fish gelatine used as carrier for vitamin or carotenoid preparations, fish gelatine or singlass used as fining ag ent in beer and wine.
(3) Except fully refined soybean oil and fat, natural mixed tocopheols (E306), natural D-alpha tocopherol, natural D-alpha tocopherol acetate, natural D-alpha tocopherol succinate from soybean sources;vegetable oils derived phytosterols and phytosterolesters from soybean sources; plant stanol es ter producted from vegetable oil sterols from soybean sources.
(4) Except when used for making distillates or ethyl alcohol of agricultural origin for spirit drinks and other alcoholic beverages, lac tito.
(5) Almond (Amydalus communis L.) hazelnuts (Corylus avellana), walnut (Juglans regia), cashew (Anacardium occidentale), pecan nuts (Carya illinaiesis), brazil nut (Bertholletia excelsa), pistachio nut (Pistacia vera), macadamia nut and queensland nut
(Macadamia terniflora) and products thereof, except nuts used for making distillates or ethyl alcohol of agricultural origin for spirit drinks and other alcoholic beveragesIn accordance with the directive 1169/2011 EC.
- GMO Declaration
Present GM-Origin Cotton NO Not-Applicable Maize NO Not-Applicable Potato NO Not-Applicable Rape NO Not-Applicable Soya NO Not-Applicable Sugarbeet NO Not-Applicable Tomato NO Not-Applicable Wheat NO Not-Applicable The following ingredients produced from genetically modified organisms are present in the product: None
In accordance with the regulation (EC) No 1829/2003 and (EC) No 1830/2003 of the European Parliament
| Value | Units | Test Method / Conditions | |
| Lead Content | max. 0.5 | ppm | ICP-MS |
| Arsenic Content | max. 0.02 | ppm | ICP-MS |
| Cadmium Content | max. 0.5 | ppm | ICP-MS |
| Mercury Content | max. 0.1 | ppm | ICP-MS |
| Value | Units | Test Method / Conditions | |
| Particle Size through 200 mesh | 100 | % | ph. EUR. / USP |
| Bulk Density | 0.4 - 0.4 | g/ml | ph. EUR. / USP |
| Loss of Drying | max. 5 | % | ph. EUR. / USP |
| Ash Content | max. 5 | % | ph. EUR. / USP |
| Value | Units | Test Method / Conditions | |
| Total Plate Count | max. 10000 | CFU/g | ph. EUR. / USP |
| Yeast and Molds Count | max. 100 | CFU/g | ph. EUR. / USP |
| Enterobacteriaceae | max. 100 | CFU/g | ph. EUR. / USP |
| Escherichia coli | Absencce / 1g | — | ph. EUR. / USP |
| Escherichia Coli | Absent | 1g | ph. EUR. / USP |
| Salmonella | Absence / 10g | — | ph. EUR. / USP |
| Salmonella | Absent | 10g | ph. EUR. / USP |
Regulatory & Compliance
- Statements
Non Irradiation/Non Ionized Statement
We hereby certify that the product has not been sterilized byionizing radiation at anypoint during the entire manufacturing process and therefore is in full compliance with the relevant legislated regulations (Directive 1999/2/CE).
BSE/TSE Statement
We hereby certify that the product contains no ingredients of ruminant origin and no materials derived from, or exposed to ruminants affected byor under quarantine for Transmitting Transmissible Spongiform Encephalopathy(TSE)/ Bovine Spongiform Encephalopathy (BSĖ) and it is conform to the EU legislation 999/2001.
Packaging Statement
We hereby certify that the packing used is in compliance with:
- Commission Regulation (EC)No 1935/2004 and Regulation (EU) No 10/2011 regarding packaging materials and articles intended to come into contact with food.
Pesticide Statement
We hereby certify, basis on our actual knowledge of production process, raw materials and equipment used potential pesticide residues in the above-mentioned product comply with the European legislation on pesticide residues, esp. Regulation (EC) No. 396/2005.
Residual Solvent Statement
We hereby certify that the product meets with:
- Directive E 2010/59/EU of 26 August 2010 amending Directive 2009/32/EC ofthe European Parliamentand of the Council on the approximation of the laws of the Member States on extraction solvents used in the production of foodstuffs and food ingredients.
Contaminant Statement
This is to certify that the product meets with:
- REGULATION (EC) No. 1881/2006 and subsequent amendments as concerns the maximum level admitted of the
Following contaminants:
- Aflatoxin B1
- Aflatoxins B1 + B2 + G1 + G2
- Ochratoxin
- Melamine
REGULATION (EC) No. 1933/2015 amending regulation (EC) No. 1881/2006 as regards maximum levels for polycyclic arom atic hydrocarbons, in particular:
- Maximum level of 10 ug/kg of benzo(a)pyrene
- 50 ug/kg for the sum of PAH4 (benzo(a)pyrene, chrysene, benz(a)anthracene and benzo(b)fluoranthene) in food supplements.
Nanomaterial Statement
According to the definition of “nanomaterials" of the EU Regulation (EU) No. 1169/2011 of the European parliament and of the council of 25 October 2011 on the provision of food information to consumers, we hereby attest that no nanomaterials are used in the formulation or in the packaging material of the product.
Anti-Doping Statement
As per the list of 2017 and subsequent amendments of the world anti-doping agency, the ingredient is not a doping substance and not a combination of doping substances. The ingredient does not contain any doping substance. The ingredient does not result from a doping substance.
Expected Usage
The expected usage ofthis product is its incorporation as an ingredientin the food industryor the pharmaceutical industry.
Technical Details & Test Data
- Plant Information
- Scientific name : Larix sibirica Ledeb.
- Synonyms : Larix russica (Endl.) Sabine ex Trautv.
Larix sukaczewii Dylis - Botanical family : Pinaceae
- Common names : Siberian larch, Russian larch
- Geographical origin : Russian Federation, Siberia
Harvest
Period (month/s or season) Winter Method (manual/mechanical) Mechanical Used part Larch Wood (butt) Type of culture (wild/cultivated) Wild After Harvest
Crushed Yes Cleaned Yes Dried Electric oven Additional Information
Treatments before/after harvest No / No Storage In dry and cool well-ventilated place away from light - Extraction
Extraction method Solvent extraction Solvent(s) 80% Ethanol / 20% Water Carrier/ excipient No Ratio 50 - 58 : 1 Active Compounds
Parameters Specification Testing Method Dihydroquercetin (Taxifolin) >(90%; 92%; 94%; 96%; 98%) HPLC (2R,3R)-Dihydroquercetin ( in dry Dihydroquercetin) >95% HPLC - Product Flow Chart
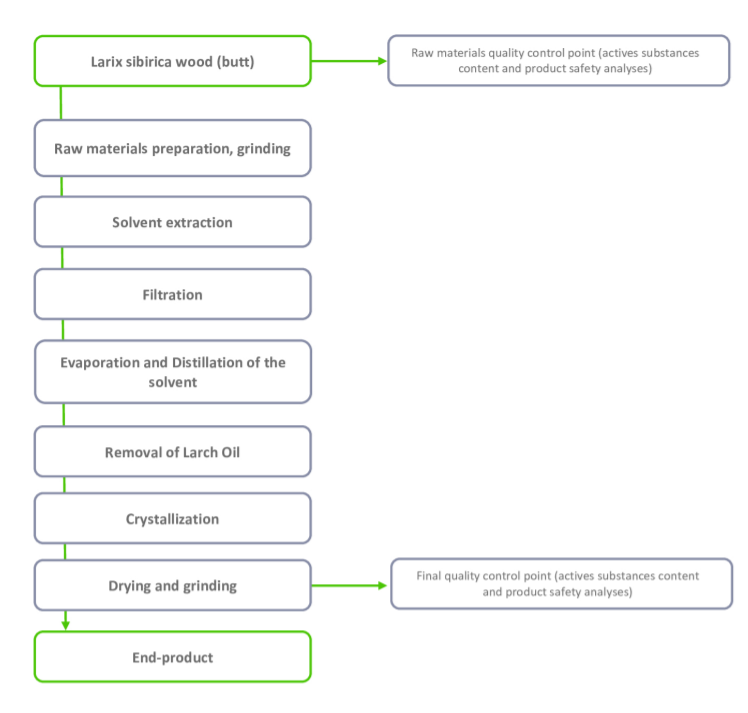
Packaging & Availability
- Packaging Information
10 kg/carton box
Storage & Handling
- Storage & Shelf Life
Stored in a well-closed container in cool and dry place, keep away from direct strong light and heat. Not less than 3 years when properly stored (see COA).