Knowde Enhanced TDS
Identification & Functionality
- Ingredient Name
- Ingredient Origin
- Food Ingredients Functions
- Ingredients
- Maltodextrin, Sea Buckthorn Dry Extract
Features & Benefits
- Benefit Claims (Health)
- Food Ingredients Features
Applications & Uses
- Markets
- Applications
- Food & Nutrition Applications
Properties
- Flavor
- Characteristic
- Typical Properties
- Physico-Chemical Properties
- Microbiological Values
- Plant Information
- Scientific name : Hippophae rhamnoides L.
- Botanical family : Elaeagnaceae
- Common names : Sea buckthorn
- Geographical origin : Russian Federation, Siberia
Harvest
Period (month/s or season) Autumn Method (manual/mechanical) Manual Used part Fruits Type of culture (wild/cultivated) Wild Plant growth stage On the ripening phase After Harvest
Crushed Yes Cleaned Yes Dried No Additional Information
Treatments before/after harvest No / No Identification Botanical Storage In dry and cool well-ventilated place away from light - Allergen Declaration EU
Allergens Direct Incorporation Cross Contamination Presence on the production line Presence on the production workshop Presence on the production factory Cereals containing gluten (1) and
products thereofNO NO NO NO Crustaceans and products
there ofNO NO NO NO Eggs and products thereof NO NO NO NO Fish and products thereof (2) NO NO NO NO Peanuts and products there of NO NO NO NO Soybeans and products there of
(3)NO NO NO NO Milk and products thereof
(including lactose) (4)NO NO NO NO Nuts (5) or products thereof NO NO NO NO Celery and products there of NO NO NO NO Mustard and products there of NO NO NO NO Sesame seeds and products
there ofNO NO NO NO Sulfur dioxide and sulphites at
concentrations of more than 10
mg/kg or 10 mg/l expressed as
SO2NO NO NO NO Lupines and products thereof NO NO NO NO Mollusk and product thereof NO NO NO NO (1) Cereals which contain gluten (i.e.w heat, rye, barley, oats, spelt, kamut or their hybridized strains) except: w heat-based glucose syrups including dextrose, wheat-based maltodextrins, glucose syrups based on barley, cereals used for making distillates or ethyl alcohol of agricultural origin for spirit drinks and other alcoholic beverages.
(2) Except: fish gelatine used as carrier for vitamin or carotenoid preparations, fish gelatine or singlass used as fining ag ent in beer and wine.
(3) Except fully refined soybean oil and fat, natural mixed tocopheols (E306), natural D-alpha tocopherol, natural D-alpha tocopherol acetate, natural D-alpha tocopherol succinate from soybean sources;vegetable oils derived phytosterols and phytosterolesters from soybean sources; plant stanol es ter producted from vegetable oil sterols from soybean sources.
(4) Except when used for making distillates or ethyl alcohol of agricultural origin for spirit drinks and other alcoholic beverages, lac tito.
(5) Almond (Amydalus communis L.) hazelnuts (Corylus avellana), walnut (Juglans regia), cashew (Anacardium occidentale), pecan nuts (Carya illinaiesis), brazil nut (Bertholletia excelsa), pistachio nut (Pistacia vera), macadamia nut and queensland nut
(Macadamia terniflora) and products thereof, except nuts used for making distillates or ethyl alcohol of agricultural origin for spirit drinks and other alcoholic beveragesIn accordance with the directive 1169/2011 EC.
- GMO Declaration
Present GM Origin Cotton NO Not-Applicable Maize YES(1) Not-GMO Potato NO Not-Applicable Rape NO Not-Applicable Soya NO Not-Applicable Sugarbeet NO Not-Applicable Tomato NO Not-Applicable Wheat NO Not-Applicable (1): From Maltodextrin
| Value | Units | Test Method / Conditions | |
| Heavy Metals Content | max. 10 | ppm | ICP-MS |
| Organic Acids (Malic acid) | min. 10 | % | Titration |
| Lead Content | max. 3 | ppm | ICP-MS |
| Carotenoids (β-carotin) | min. 10 | mg/100g | UV-VIS |
| Arsenic Content | max. 2 | ppm | ICP-MS |
| Cadmium Content | max. 1 | ppm | ICP-MS |
| Mercury Content | max. 0.1 | ppm | ICP-MS |
| Value | Units | Test Method / Conditions | |
| Particle Size through 35 mesh | 100 | % | ph. EUR. / USP |
| Bulk Density | 0.4 - 0.7 | g/ml | ph. EUR. / USP |
| Loss of Drying | max. 5 | % | ph. EUR. / USP |
| Ash Content | max. 5 | % | ph. EUR. / USP |
| Value | Units | Test Method / Conditions | |
| Total Plate Count | max. 50000 | CFU/g | ph. EUR. / USP |
| Yeast and Molds Count | max. 500 | CFU/g | ph. EUR. / USP |
| Enterobacteriaceae | max. 100 | CFU/g | ph. EUR. / USP |
| Escherichia coli | Absence / 1g | — | ph. EUR. / USP |
| Escherichia Coli | Absent | 1g | ph. EUR. / USP |
| Salmonella | Absence / 25g | — | ph. EUR. / USP |
| Salmonella | Absent | 25/g | ph. EUR. / USP |
Regulatory & Compliance
- Statements
Non Irradiation/Non Ionized Statement
We hereby certify that the product has not been sterilized byionizing radiation at anypoint during the entire manufacturing process and therefore is in full compliance with the relevant legislated regulations (Directive 1999/2/CE).
BSE/TSE Statement
We hereby certify that the product contains no ingredients of ruminant origin and no materials derived from, or exposed to ruminants affected byor under quarantine for Transmitting Transmissible Spongiform Encephalopathy(TSE)/ Bovine Spongiform Encephalopathy (BSĖ) and it is conform to the EU legislation 999/2001.
Packaging Statement
We hereby certify that the packing used is in compliance with:
- Commission Regulation (EC)No 1935/2004 and Regulation (EU) No 10/2011 regarding packaging materials and articles intended to come into contact with food.
Pesticide Statement
We hereby certify, basis on our actual knowledge of production process, raw materials and equipment used potential pesticide residues in the above-mentioned product comply with the European legislation on pesticide residues, esp. Regulation (EC) No. 396/2005.
Residual Solvent Statement
We hereby certify that the product meets with:
- Directive E 2010/59/EU of 26 August 2010 amending Directive 2009/32/EC ofthe European Parliamentand of the Council on the approximation of the laws of the Member States on extraction solvents used in the production of foodstuffs and food ingredients.
Contaminant Statement
This is to certify that the product meets with:
- REGULATION (EC) No. 1881/2006 and subsequent amendments as concerns the maximum level admitted of the
Following contaminants:
- Aflatoxin B1
- Aflatoxins B1 + B2 + G1 + G2
- Ochratoxin
- Melamine
REGULATION (EC) No. 1933/2015 amending regulation (EC) No. 1881/2006 as regards maximum levels for polycyclic arom atic hydrocarbons, in particular:
- Maximum level of 10 ug/kg of benzo(a)pyrene
- 50 ug/kg for the sum of PAH4 (benzo(a)pyrene, chrysene, benz(a)anthracene and benzo(b)fluoranthene) in food supplements.
Nanomaterial Statement
According to the definition of “nanomaterials" of the EU Regulation (EU) No. 1169/2011 of the European parliament and of the council of 25 October 2011 on the provision of food information to consumers, we hereby attest that no nanomaterials are used in the formulation or in the packaging material of the product.
Anti-Doping Statement
As per the list of 2017 and subsequent amendments of the world anti-doping agency, the ingredient is not a doping substance and not a combination of doping substances. The ingredient does not contain any doping substance. The ingredient does not result from a doping substance.
Expected Usage
The expected usage ofthis product is its incorporation as an ingredientin the food industryor the pharmaceutical industry.
Technical Details & Test Data
- Extraction
Extraction method Solvent extraction Solvent(s) Water Drying method Vacuum impulse Carrier/ excipient 20-28% maltodextrin Ratio 10:1 - Product Flow Chart
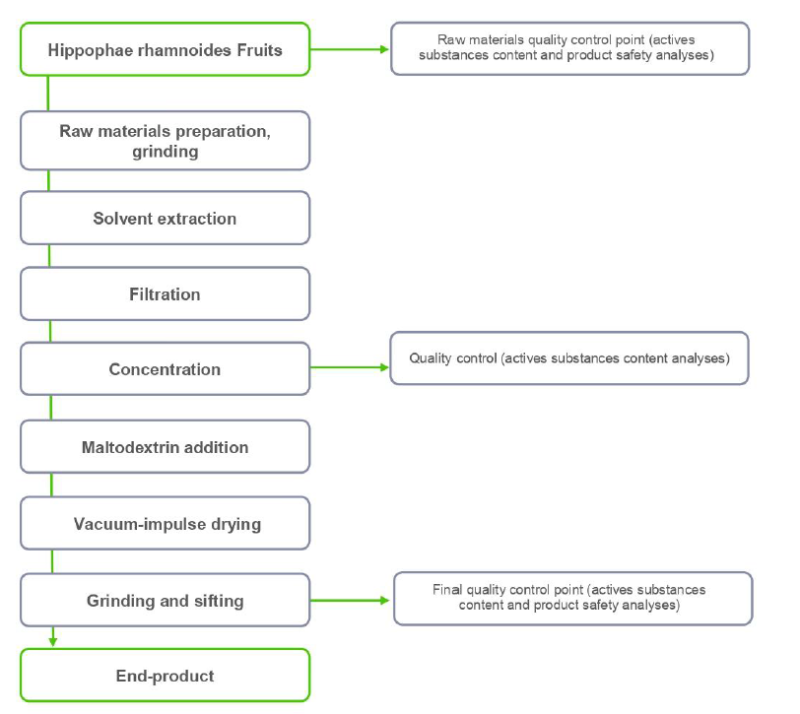
Packaging & Availability
- Packaging Information
10 kg/carton box
Storage & Handling
- Storage & Shelf Life
Stored in a well-closed container in cool and dry place, keep away from direct strong light and heat. Not less than 3 years when properly stored (see COA).