Knowde Enhanced TDS
Identification & Functionality
- Chemical Name
- Pharma & Nutraceuticals Functions
- CAS No.
- 303-98-0
- EC No.
- 206-147-9
- Technologies
- Product Families
Applications & Uses
- Markets
- Dosage Form
- Use application
For beverage (water-based drinks)
Properties
- Appearance
- off white - fine yellow
- Typical Properties
| Value | Units | Test Method / Conditions | |
| Content of Ubiquinol | max. 40.0 | % | HPLC method |
| Heavy metal | min. 10 | μg/g | Food Sanitation Law(JP) |
| Number of live bacteria | min. 300 | cfu/g | Standard Methods of Analysis in Food Safety Regulations(JP) |
Regulatory & Compliance
- Certifications & Compliance
- Grade
Technical Details & Test Data
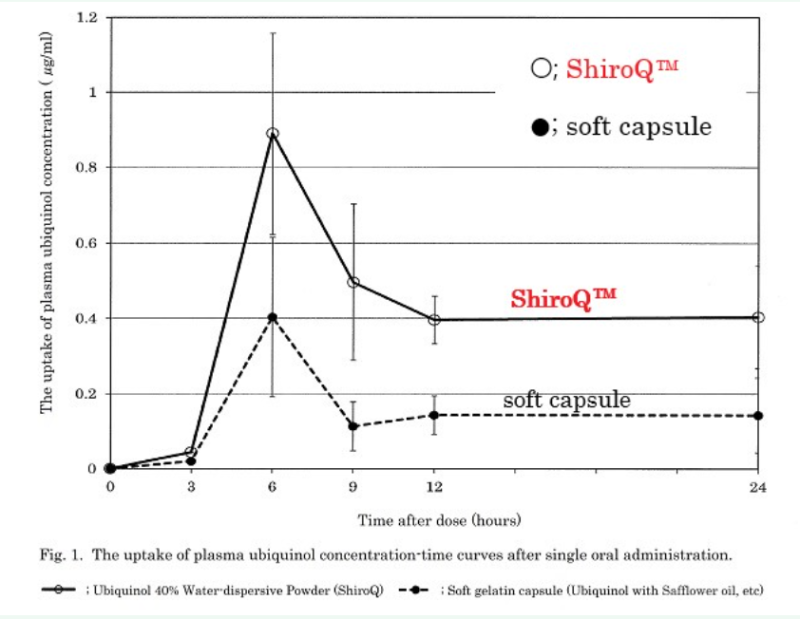
- The bioavailability of reduced coenzyme Q10 Water-dispersive Powder(ShiroQ) after single oral administration.
In this study, we showed the bioavailability of ubiquinol (QH) in the form of Water-dispersive Powder(ShiroQ). Two groups of 5 healthy young subjects received single oral administration of 100mg of QH in the form of a soft capsule containing QH dissolved in safflower oil or 40% Water-dispersive Powder(ShiroQ) in the fasting period, and changes in the plasma QH concentration were monitored over time. The plasma QH concentration before administration was 0.68 ± 0.08 μg/ml. Tmax was 6 hours after the administration. Cmax values compared with the pre-administration baseline in the soft capsule and ShiroQ groups were 0.4 ± 0.21 and 0.89 ± 0.27 μg/ml, respectively, and AUC0-24hr values were 3.59 ± 1.63 and 9.68 ± 2.35 μg·h/ml, respectively. The Water-dispersive Powder form of QH (ShiroQ) exhibited superior bioavailability even when administered in the fasting period.