Knowde Enhanced TDS
Identification & Functionality
- Chemical Name
- Pharma & Nutraceuticals Functions
- CAS No.
- 74811-65-7
- EC No.
- 629-739-2
- Technologies
Features & Benefits
- Labeling Claims
- Benefits
- Rapid disintegration
- Aid in dissolution
- Ensuring the bioavailability of active ingredient
- No effect on the compressibility and flowability
- Cost-effective at low use level
- Product Highlights
Meets the Requirement for Pharmaceutical and Food applications
Disolcel® complies with the monograph requirements of current edition of the pharmacopeias:USP/NF, EP, BP and JP.
Applications & Uses
- Applications
Properties
- Typical Properties
| Value | Units | Test Method / Conditions | |
| Loss on Drying | max. 10.0 | % | — |
| Settling Volume | '10-30 | ml | — |
| Degree of Substitution | 0.60 - 0.85 | — | — |
Regulatory & Compliance
- Certifications & Compliance
- Grade
Technical Details & Test Data
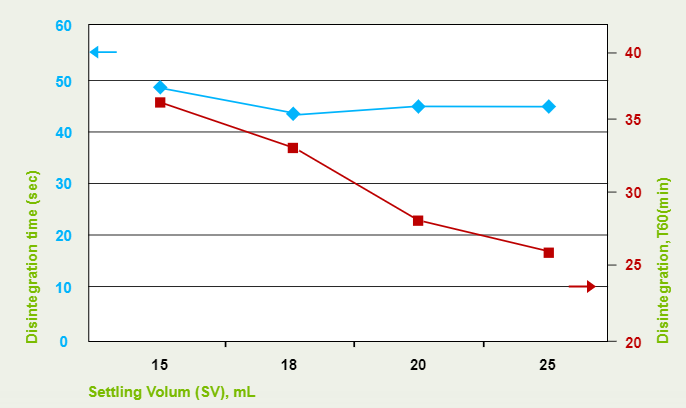
- Effect of Settling Volumes in Disolcel® on Drug Dissolution
The data below shows that formulation containing Disolcel® with higher settling volume, has faster drug dissolution that means speed up the drug released rate. (i.e the shorter T60%)