Knowde Enhanced TDS
Identification & Functionality
- Active Component
- Chemical Family
- Ingredient Name
- Ingredient Origin
- Food Ingredients Functions
- Pharma & Nutraceuticals Functions
- Ingredients
- Policosanol
- Product Families
- Labeling Suitability
- Suitable for Vegans, Vegetarians, and Diabetics
Features & Benefits
- Labeling Claims
- Food Ingredients Features
- Benefits
The research of Dr. Cureton from the University of Illinois, U.S.A. reported the usefulness of natural vegetable waxes to our health. Additional research has been conducted in Cuba and elsewhere establishing the efficacy of higher aliphatic alcohols in the human diet. 1-Octacosanol is the most prolific aliphatic alcohol in Policosanol.
- Advantages
- Unlike other ingredient forms of aliphatic fatty alcohols mixtures such as those derived from rice bran wax, bees wax, wheat germ and other plant and insect waxes, LesstanoL® policosanol (polycosanol) raw material is derived solely from pure sugarcane wax, the only source of fatty alcohols with a large volume of published clinical studies showing its effectiveness.
- This is most likely because sugarcane wax derived aliphatic fatty alcohols as a raw material, have the highest content of 1-octacosanol and it has very specific ratios of aliphatic fatty alcohols that are not found in rice bran wax, wheat germ wax, bees wax or other types of raw material waxes. Rice bran wax, wheat germ wax, bees wax and other waxes may not be as effective an ingredient at supporting health.
- Quality Assurance
Full quality assays are done on every batch of LesstanoL® Policosanol (polycosanol) raw material by an independent state certified testing laboratory before the batch is released from Quality Control. Ingredient analyzes conducted include full fatty alcohols profile, heavy metals profile, full microbiological profile and periodically, full pesticide residue screening. An analysis certificate accompanies each shipment of this raw material to assure you that you are receiving the finest quality ingredient available.
Applications & Uses
- Applications
- Food & Nutrition Applications
- Applications
- Supplementation: LesstanoL® brand Policosanol (polycosanol) and Octacosanol raw material can be used as an ingredient to prepare various types of dietary supplement tablets or capsules. Most manufacturer’s purchasing this ingredient, formulate it in dietary supplements designed to support health. Because Policosanol is oil soluble and used in such small concentrations (5-10 mg/serving), this ingredient can also be added to high fat systems such as vegetables oils, margarine, salad dressing, nut butters and spreads.
- Beverage uses: LesstanoL® brand Policosanol and Octacosanol can be used to formulate sports-type, electrolyte replacement and carbohydrate-loading beverages. Various value-added, health-oriented beverages can be prepared using LesstanoL®Octacosanol in combination with an emulsifier.
Properties
- Solubility
- Appearance
- Fine granular powder, white to beige
- Chemical Properties
- Microbiological Values
- Note
- *Based on minimum detection level of < 3 MPN/g.
- **Based on minimum detection level of < 10 CFU/g.
| Value | Units | Test Method / Conditions | |
| 1-Dotriacontanol | 0.1 - 10 | % | GC-FID (Proprietary method) |
| 1-Heptacosanol | 0 - 2.0 | % | GC-FID (Proprietary method) |
| 1-Hexacosanol | 2 - 15 | % | GC-FID (Proprietary method) |
| 1-Nonacosanol | 0 - 10 | % | GC-FID (Proprietary method) |
| 1-Octacosanol | 55 - 70 | % | GC-FID (Proprietary method) |
| 1-Tetracosanol | 0 - 10 | % | GC-FID (Proprietary method) |
| 1-Tetratriacontanol | 0.1 - 10 | % | GC-FID (Proprietary method) |
| 1-Triacontanol | 5 - 24 | % | GC-FID (Proprietary method) |
| Arsenic Content (As) | max. 1 | ppm | AOAC 993.14 (Modified) |
| Copper Content (Cu) | max. 10 | ppm | AOAC 993.14 (Modified) |
| Irradiation | Not Irradiated | — | — |
| Lead Content (Pb) | max. 2 | ppm | AOAC 993.14 (Modified) |
| Loss on Drying | max. 1.0 | % | ASTA 2.1 |
| Mercury Content (Hg) | max. 1 | ppm | AOAC 993.14 (Modified) |
| Pesticides (or) Herbicides (Specified list) | Less than detection limits | — | FDA – Pam/Luke – /GC-MS |
| Primary Alcohols Wet Basis (Policosanol) | min. 90 | % | GC-FID (Proprietary method) |
| Value | Units | Test Method / Conditions | |
| Coliform | max. 10 | MPN/g | AOAC 966.23 |
| E. coli | Negative* | MPN/g | AOAC 966.23 |
| Mold | max. 100 | CFU/g | APHA CMMEF CHP 21 |
| Salmonella | Negative | per 25g | AOAC 2011.03 |
| Staphylococcus aureus | Negative** | CFU/g | FDA BAM ONLINE CHP 12 |
| Total Aerobic Plate Count | max. 1,000 | CFU/g | APHA CMMEF CHP 8 |
| Yeast | max. 100 | CFU/g | APHA CMMEF CHP 21 |
Regulatory & Compliance
- Certifications & Compliance
Technical Details & Test Data
- A comparison of LesstanoL® Policosanol and other popular sources of 1-Octacosanol for % of 1-Octacosanol.
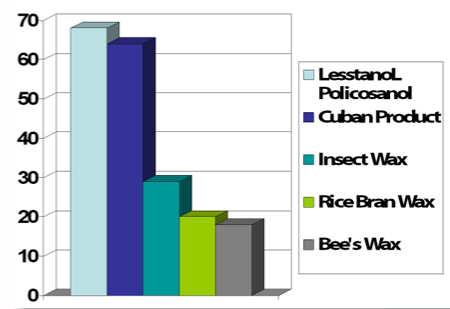
- Manufacturing Method
- Extraction, purification and distillation of sugar cane wax, no excipients or preservatives
Packaging & Availability
- Packaging
- Packed in multi-walled poly-barrier bags containing 1 kg, 5 kg and 10 kg net weights. Bags are packed in cardboard boxes or fiber drums.
Storage & Handling
- Shelf Life
- 5 years
- Storage Conditions
- Store in a cool dry location (5°- 20° C., < 60% R.H.) in an area with fresh, clean, circulating air. Protect from direct sunlight, microbial and other contamination.
- Shelf Life
- Minimum 5 years in original sealed package under suggested storage conditions

