Knowde Enhanced TDS
Identification & Functionality
- Active Component
- Ingredient Name
- Ingredients
- Olive Oil, Arabica Gum, CBD Powder
- Product Families
- Composition
20% active cannabinoid load; all natural, clean-label carriers and non- synthetic emulsifiers
- Derived from
Pure isolates or refined hemp distillates derived from whole hemp plants
Features & Benefits
- Labeling Claims
- Food Ingredients Features
Applications & Uses
- Markets
- Food & Nutrition Applications
- Applications
- Water-soluble powder is ideal for ready-to-drink mixes, stick packs, bulk powder tubs, shots, foods, beverages and supplements.
- Each application is very different : The stability and performance of the cannabinoid ingredients are highly dependent on the other ingredients in the product. Factors like pH, sugar content, manufacturing conditions and container type and size can all play a major role in flavor and stability.
Properties
- Solubility
- Appearance
- Fine, free-flowing, white powder
- Flavor
- Neutral to slightly bitter
- Odor
- Neutral to faint terpenoid
- Soluble in
- Water
- Typical Properties
- Microbiological Values
- Heavy Metals
- Cannabinoid Content
- Note
* This analysis or component is not ISO accredited.
| Value | Units | Test Method / Conditions | |
| Moisture Content* | 2.0 | % | Karl Fischer method |
| Consistency | Fine powder | - | - |
| Value | Units | Test Method / Conditions | |
| Aerobic Plate Count* | max. 100 | cfu/g | - |
| Escherichia Coli* | Absent | per 10g | - |
| Enterobacterial Count* | max. 10 | MPN/g | - |
| Yeast Count* | max. 100 | cfu/g | - |
| Mold Count* | max. 100 | cfu/g | - |
| Value | Units | Test Method / Conditions | |
| Arsenic Content | max. 10.2 | ppb | Dry Basis |
| Cadmium Content | max. 5.1 | ppb | Dry Basis |
| Lead Content | 13.5 | ppb | Dry Basis |
| Mercury Content | max. 5.1 | ppb | Dry Basis |
| Value | Units | Test Method / Conditions | |
| CBDVA | max. 0.00255 | % | Dry Basis |
| CBDV | 0.0424 | % | Dry Basis |
| CBDa | max. 0.00255 | % | Dry Basis |
| CBGa | max. 0.00255 | % | Dry Basis |
| CBG | max. 0.00255 | % | Dry Basis |
| CBD | 21.7 | % | Dry Basis |
| THCV | max. 0.00255 | % | Dry Basis |
| CBN | max. 0.00255 | % | Dry Basis |
| Δ9-THC | max. 0.00255 | % | Dry Basis |
| Δ8-THC | max. 0.00510 | % | Dry Basis |
| THCA | max. 0.00255 | % | Dry Basis |
| CBC | max. 0.00255 | % | Dry Basis |
| THCVA | max. 0.00255 | % | Dry Basis |
| CBNA | max. 0.00255 | % | Dry Basis |
| CBCA | max. 0.00255 | % | Dry Basis |
| CBL | max. 0.00255 | % | Dry Basis |
| Total Cannabinoids | 21.8 | % | Dry Basis |
| Total THC (THC + (THCA x 0.877)) | max. 0.00255 | % | Dry Basis |
| Total CBD (CBD + (CBDA x 0.877)) | 21.7 | % | Dry Basis |
Regulatory & Compliance
- Compliance
- United Kingdom: Novel Foods compliance in process
- European Union: Novel Foods compliance in process
- Produced in cGMP facilities
- Validated using trusted third-party accredited laboratories
Technical Details & Test Data
- Water-soluble Powder Has Demonstrably Better Bioavailability
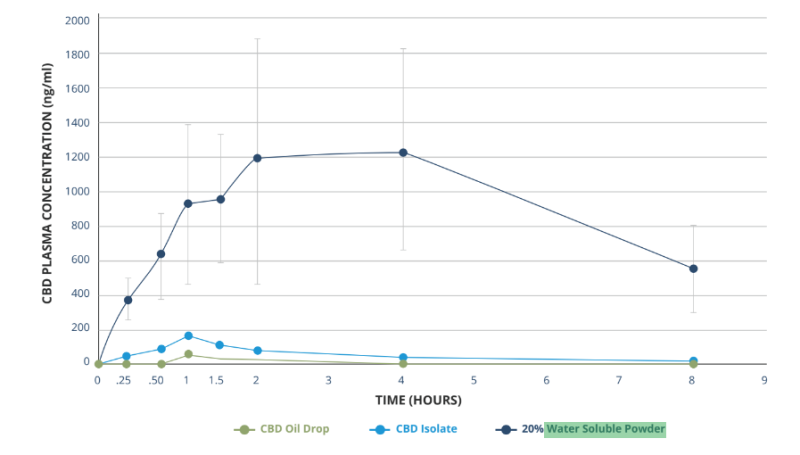
Plasma pharmacokinetic (PK) study in rats following single oral administration of three (3) cannabidiol formulae
As shown in the graph, this product format is more bioavailable than cannabinoids are on their own, which allows for broad use in liquid and powder formulations - from shots and beverages to food, supplements and pet applications.
Storage & Handling
- Shelf Life
- 24 months