Knowde Enhanced TDS
Identification & Functionality
- Ingredient Origin
- Cosmetic Ingredients Functions
- Technologies
- Product Families
- Chemical Structure
- Origin
- Hydrolyzed Trigonella Foenum-Graecum Seed Extract - Vegetable
Features & Benefits
- Benefit Claims
- Labeling Claims
- Action
- Anti-aging, delays the aging process
- Reduces wrinkles and lines
- Improves elasticity and firmness
- Increases epidermal thickness
- Reinforces the skin barrier
- Cosmetic Benefits
- The whole process and the signs of aging are delayed.
- The epidermis regains thickness and firmness, skin is more elastic and supple, lines and wrinkles fade.
Applications & Uses
- Application Format
- Skin Care Applications
- Cosmetic Use
- Anti-aging care products
- Anti-wrinkle care products
- Firming care products
- Regenerative care products
- Moisturizing care products
- Formulations
- Concentration for use: 1%
- pH for use: 4.0 to 10.0
- Caution for use: Solubilize previously in the aqueous phase. Add to emulsions, at the end of the preparation process, either cold or at 35-40 °C, during cooling.
Properties
- Odor
- Characteristic
- Appearance
- Powder
- Insoluble in
- 10% in ethanol
- Partially Soluble in
- 10% in water under stirring (500 rpm)
- Typical Properties
- Quantifications
- Physico-Chemical Properties
- Microbiological Values
| Value | Units | Test Method / Conditions | |
| Natural Index | 0 | — | — |
| Average Molecular Weight | max. 20 | kDa | — |
| Natural Origin Index | 0 | — | — |
| Value | Units | Test Method / Conditions | |
| Granulometry (<1mm) | min. 80 | % | GT039 |
| Total sugars (exp. eq. mannose-galactose) | min. 80 | %/MS - %/DM | GT044 |
| Value | Units | Test Method / Conditions | |
| Humidity | max. 15.0 | % | GT006 |
| pH Value (Solution 1% w/w in Water) | 4.0 - 6.5 | — | GT005 |
| Value | Units | Test Method / Conditions | |
| Detection of Enterobacteria | Absence | — | ISO 18415 |
| Enumeration of Mesophilic Aerobic Bacteria | max. 100 | ufc/g - cfu/g | ISO 21149 |
| Enumeration of Yeasts and Molds | max. 100 | ufc/g - cfu/g | ISO 16212 |
Regulatory & Compliance
- Certifications & Compliance
- REACH Status
INCI Concentration REACH Status Hydrolyzed Trigonella Foenumraecum
Seed Extract100 % Exempt: < 1 ton/year (dry matter)
Technical Details & Test Data
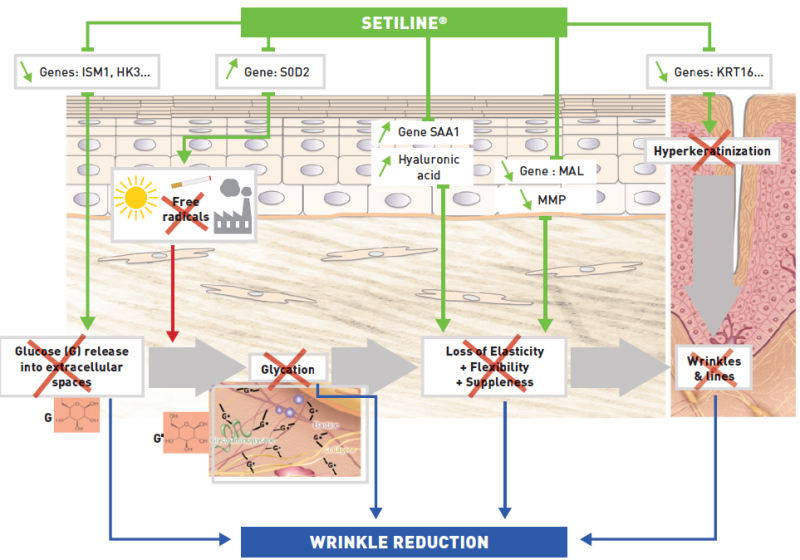
- Mechanism of Action
SETILINE® improves the skin overall architecture, by preserving it from dermal collagen and integrin glycation phenomena (major cause of aging) and strengthens the skin barrier by reducing keratinocyte differentiation.
- Scientific Process
SETILINE® modifies the expression of specific genes involved in the glycation of collagens and integrins in the dermis, thereby maintaining a solid and flexible skin structure. This active substance inhibits the build-up of advanced glycation end-products (AGEs) and displays properties similar to those of retinoic acid by stimulating involucrin synthesis.
SETILINE® improves the biomechanical properties of the dermis and epidermis and preserves their structures, thus delaying the aging process.- DNA Chip Transcriptome Analysis
- Proven Efficacy - In Vitro Test
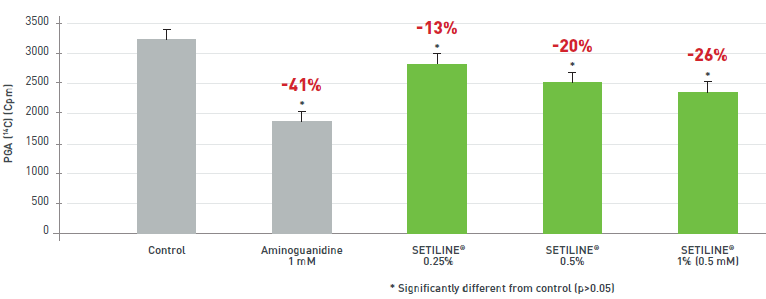
ANTI-GLYCATION: REDUCTION IN ADVANCED GLYCATION END-PRODUCTS (AGES) (DERMAL action)
- SETILINE® inhibits AGE accumulation.
- SETILINE® improves dermal flexibility and elasticity.
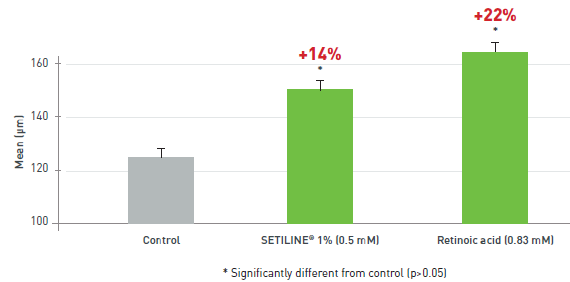
KERATINOCYTE DIFFERENTIATION (EPIDERMAL action)
- SETILINE® possesses properties similar to those of retinoic acid. SETILINE® stimulates involucrin expression and improves the skin barrier function, thus preventing aging.

EPIDERMAL THICKENING (EPIDERMAL action)
- During aging, the skin becomes thinner. SETILINE® increases epidermal thickness.
- Proven Efficacy - In Vivo Test
EVALUATION ON VOLUNTEERS*
- Cutometer® measurements (16 volunteers)
> Elasticity: + 45%, up to +214%
> Firmness: up to +93%
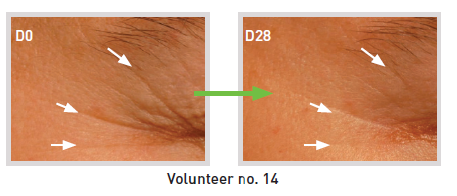
- Fringe projection measurements / Primos® compact (15 volunteers)
> Anti-wrinkle: up to -37%
> Skin micro-profile smoothing: up to -30.5%
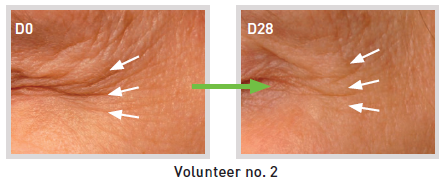
* - 28 days / 1% SETILINE® / twice-daily application
SELF-EVALUATION*
- 81% of volunteers observed a reduction in wrinkles and lines, along with smoother skin
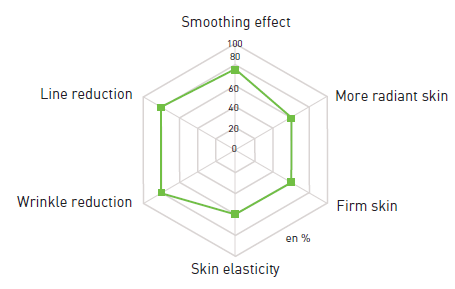
*28 days / 1% SETILINE® / twice-daily application / 21 volunteers
- Cutometer® measurements (16 volunteers)
Safety & Health
- Toxicological Tests
Tests Method Tested Concentration Results Skin Irritation Patch Test 5 % Not irritant Eye Irritation Het cam 5 % Pratically non irritant Sensitization Marzulli-Maibach 10 % Hypoallergenic Mutagenicity Test Ames test 5 % Not mutagenic
and not promutagenicPhototoxicit 3T3 cells Product tested at 8
dilutions
Storage & Handling
- Shelf Life
- 24 months
- Storage
15 / +25°C
Keep in the original packaging, without opening, in the recommended storage conditions

