Knowde Enhanced TDS
Identification & Functionality
- Carrier
- INCI Name
- Ingredient Origin
- Cosmetic Ingredients Functions
- Technologies
Features & Benefits
- Benefit Claims
- Labeling Claims
Applications & Uses
- Markets
- Application Format
- Skin Care Applications
Properties
- Appearance
- Light yellow to reddish brown capsule dispersion liquid
- Odor
- characteristic odor
- Specifications
| Value | Units | Test Method / Conditions | |
| Arsenic Content | max. 2 | ppm | - |
| Heavy Metals Content | max. 20 | ppm | - |
| Residue on Evaporation | 3.0 - 5.5 | w/v% | - |
Regulatory & Compliance
- Certifications & Compliance
Technical Details & Test Data
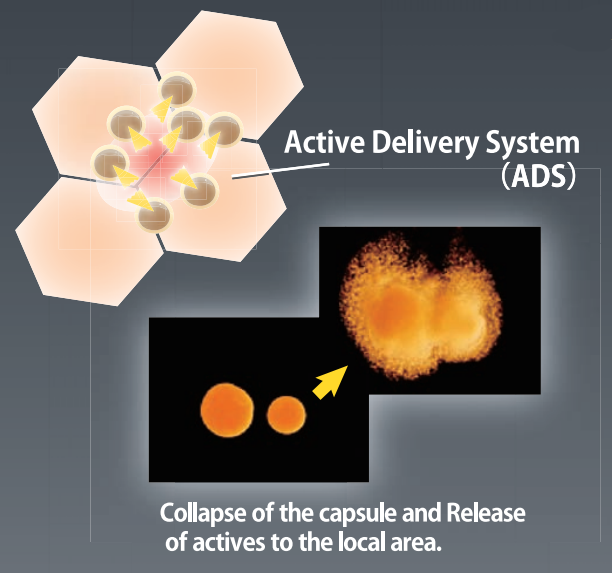
- Active Delivery System
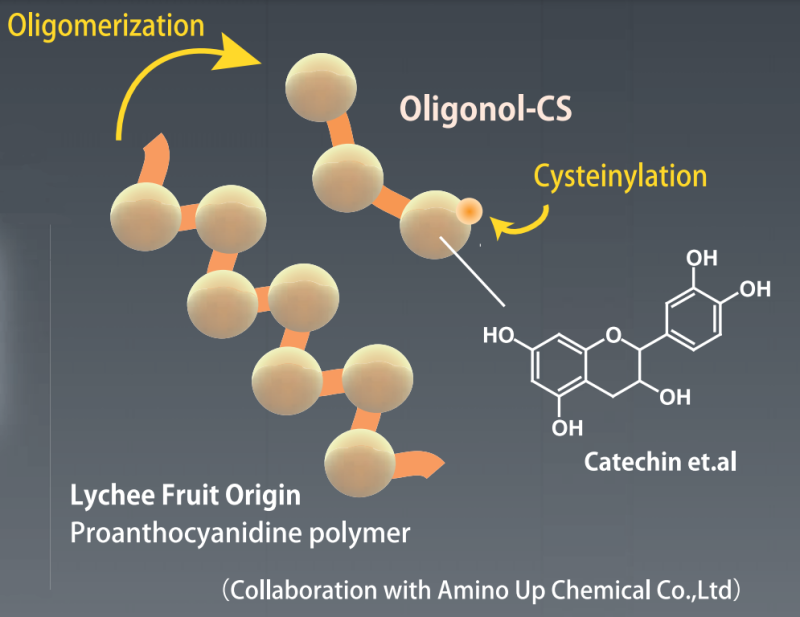
- Active Substance Oligonol CS
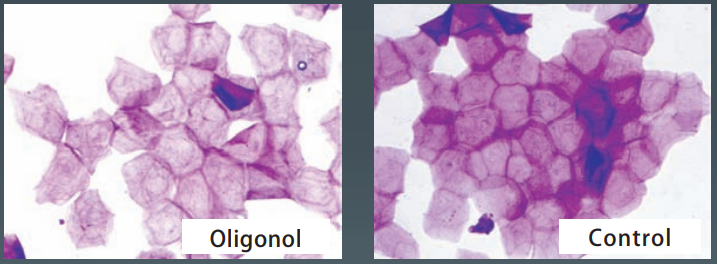
- Normalization of Stratum Corneum
After the application of lotion containing 10% of Oligonol-CS for 2 months, and tape – stripped corneocyte were collected. The corneocyte of the area that applied Oligonol-CS were in good condition with regular structure, however, the structure of stratum corneum were disordered in the control (1,3-BG) area.
- Whitening
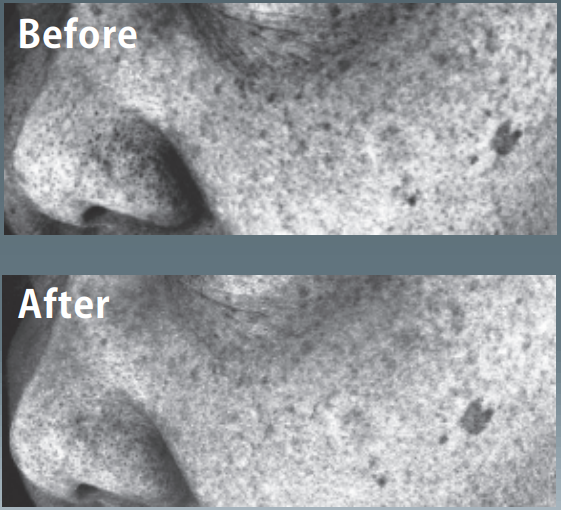
After the application of lotion containing 10% of Oligonol-CS for 2 months, decreased facial pigmentations was observed.
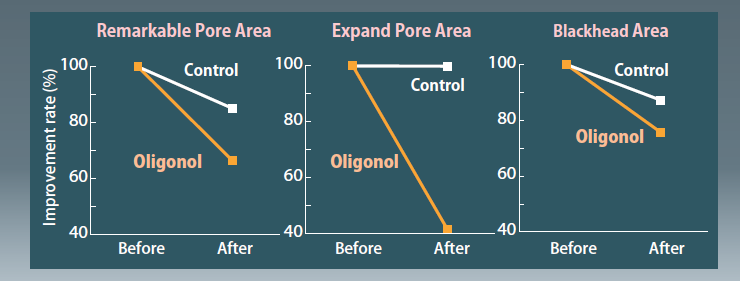
- Improvement in Pore Condition
After the application of lotion containing 10% of Oligonol-CS for 2 months, noticeable pores were virtually imperceptible.