Knowde Enhanced TDS
Identification & Functionality
- Chemical Name
- Pharma & Nutraceuticals Functions
- CAS No.
- 1397-89-3
- EC No.
- 215-742-2
- Technologies
- Product Families
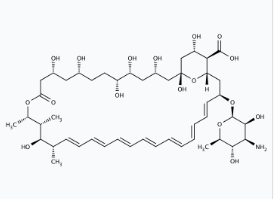
- Molecular Structure
Applications & Uses
Properties
- Appearance
- Yellow or orange powder
- Typical Properties
- Microbiological Values
- Specifications
| Value | Units | Test Method / Conditions | |
| Molecular Weight | 924.079 | — | — |
| Value | Units | Test Method / Conditions | |
| Bacterial endotoxins | max. 1 | IU/mg | — |
| Escherichia coli | Negative | — | — |
| Salmonella | Negative | /10g | — |
| Total Aerobic Count | 100 | cfu/g | — |
| Value | Units | Test Method / Conditions | |
| Acetone | max. 5000 | ppm | — |
| Amphoterecin A | max. 5 | % | Dried Basis |
| Amphoterecin B Methyl ester | max. 2 | % | — |
| Any Other Impurity | max. 2 | % | — |
| Assay (Microbial) | min. 840 | IU/mg | Dried Basis |
| Assay Content | min. 90 | % | HPLC Method |
| DMF | max. 6000 | ppm | — |
| Ethanol Content | max. 5000 | ppm | — |
| Loss on Drying | max. 5 | % | — |
| Residue on Ignition | max. 0.5 | % | — |
| Solubility | Passes test | — | — |
| Total Impurities | max. 9.0 | % | — |
| Total Unknown Impurities | max. 7 | % | — |
Regulatory & Compliance
- Certifications & Compliance
- Grade
Safety & Health
- Hazard Statements
- H315 : Causes skin irritation
- H319 : Causes serious eye irritation
- H335 : May cause respiratory irritation
- Precautionary Statements
- P261 : Avoid breathing dust/fume/gas/mist/vapors/spray
- P305+351+338 : IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses if present and easy to do, continue rinsing
Storage & Handling
- Storage Temperature
- 2 to 8°C

