Knowde Enhanced TDS
Identification & Functionality
- Chemical Name
- Pharma & Nutraceuticals Functions
- CAS No.
- 97519-39-6
- EC No.
- 810-182-0
- Technologies
- Product Families
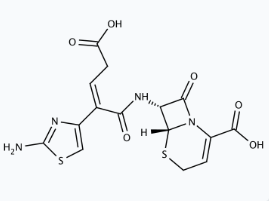
- Molecular Structure
Applications & Uses
Properties
- Appearance
- White to pale yellow crystalline powder
- Typical Properties
- Specifications
| Value | Units | Test Method / Conditions | |
| Molecular Weight | 410.42 | — | — |
| Value | Units | Test Method / Conditions | |
| Assay Content | 90.0 - 102.0 | % | Anhydrous Basis |
| Heavy Metals | max. 10 | ppm | — |
| Residue on Ignition | max. 0.1 | % | — |
| Single Impurity | max. 1 | % | — |
| Specific Optical Rotation | 135 - 155 | Degree | — |
| Total Impurities | max. 2 | % | — |
| Water Content | 8.0 - 13.0 | % | — |
Regulatory & Compliance
- Certifications & Compliance
Safety & Health
- Hazard Statements
- H332 : Harmful if inhaled
- H334 : May cause allergy or asthma symptoms or breathing difficulties if inhaled
- Precautionary Statements
- P260 : Do not breathe dust/fume/gas/mist/vapors/spray
- P280 : Wear protective gloves/protective clothing/eye protection/face protection
- P304+340 : IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for breathing
- P308+313 : IF exposed or concerned: Get medical advice/attention
Storage & Handling
- Storage Temperature
- -15 to -20°C

