Knowde Enhanced TDS
Identification & Functionality
- Chemical Family
- Chemical Name
- Pharma & Nutraceuticals Functions
- CAS No.
- 9004-34-6
- EC No.
- 232-674-9
- Technologies
Features & Benefits
- Labeling Claims
- Features
Product Highlights
Our special low viscosity grades offer unique advantages for tablet formulations: Excellent tablet hardness/friability with fast disintegration and active release times
Ease of use in wet granulation and coating applications Highest compressibility at low usage level in dry binding applications.- Highest efficiency tablet binder
- Soluble in water and polar organic solvents
- Nonionic, nonreactive, nontoxic
- Eight different molecular weight grades produced designed for immediate or sustained release
- Three different particle size grades tailored to wet or dry application
- Minimal lot-to-lot variability enabling Quality By Design
- Global regulatory approval in food and pharma
- 5-year shelf life
- No additives such as silica
Applications & Uses
- Applications
- Dosage Form
- Manufacturing Technology
- Application
Controlled Release Matrix Tablets (Dry / Powder Use/Wet / Solution Use)
- Image of Granulation using CELNY as Bridging Polymer
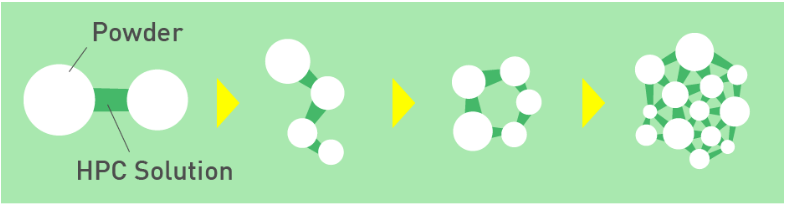
Properties
- Typical Properties
| Value | Units | Test Method / Conditions | |
| Viscosity (20℃/2%) | 4001-6000 | mPa·s | — |
| Molecular weight / GPC-RI method | 2500000 | — | — |
Packaging & Availability
- Availability
- Regular Powder (40 mesh pass) D50: 150-190 μm
- Fine Powder (100 mesh pass) D50: 80-110 μm

