Knowde Enhanced TDS
Identification & Functionality
- Backing Material Type
- Chemical Family
- Product Type
- Technologies
- Construction
- Carrier Type - Polyurethane Film
- Structure
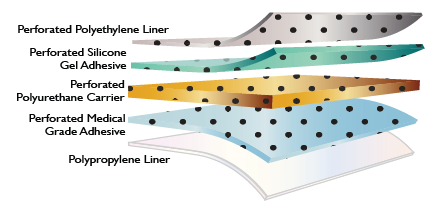
Features & Benefits
- Labeling Claims
- Ready-to-Use Product Features
- Features
- Excellent Instantaneous Tack
- Atraumatic Removal from Skin
- Hypoallergenic
- Ability to be Removed and Repositioned
- Excellent Converting Properties
Applications & Uses
- Markets
Properties
- Construction Properties
| Value | Units | Test Method / Conditions | |
| Adhesive Coat Weight | 37.0 | gsm | QSP-724 |
| Biocompatibility (Buehler Sensitization) | Pass | - | ISO 10993 |
| Biocompatibility (Cytotoxicity) | Pass | - | ISO 10993 |
| Biocompatibility (Primary Dermal) | Pass | - | ISO 10993 |
| Carrier Thickness | 25.0 | Micron | QSP-726 |
| Continuous Use Conditions | 10 - 70 | °C | QSP-754 |
| Medical Grade Adhesive Adhesion | min. 8 | N/25mm | QSP-722 |
| Perforation Hole Size/Open Area | 1.5 | mm/17% | - |
| Silicone Adhesion | 1.4 | N/25mm | QSP-723 |
| Silicone Gel Coat Weight | 150.0 | gsm | QSP-724 |
| Thickness (Excluding Liners) | 0.22 | mm | QSP-726 |
Regulatory & Compliance
- Certifications & Compliance

