Knowde Enhanced TDS
Identification & Functionality
- Chemical Family
- Chemical Name
- INCI Name
- Ingredient Origin
- Cosmetic Ingredients Functions
- CAS No.
- 23434-88-0
- EC No.
- 927-574-3
- Technologies
- Product Families
- Product Details
- Product code : 0496
- Category : Intended for Cosmeceutical application
- Botanical/Scientific name : Piper nigrum
- Plant part : Fruits
- Preparation type : Extraction
- Solvent used for extraction : Methanol
- Solvent used in manufacture : Isopropyl alcohol, Hexane and Ethanol
- Final extract ratio : 65:1
- Standardization : Tetrahydropiperine
- Excipient used : None
Features & Benefits
- Benefit Claims
- Labeling Claims
- Product Highlights
The quest for novel topical delivery systems for bioactives with emphasis on “natural” and “safe” is an area of dynamic research in the cosmeceutical industry. It has been understood that for nutrient delivery through the skin, it acts as a porous organ which like a sieve allows a certain size of minerals, fats, proteins and sugars to enter while leaving the larger ones out.
There are experiments done both in vitro and in vivo with the parent compound piperine for its bioavailability enhancing property, which indicate that Cosmoperine® may operate by increasing either of two events:
- Membrane fluidity
- Affinity of nutrient to the cell membrane
It should be also considered that Cosmoperine®, which is a lipophylic compound, may increase solubilization of the intracellular lipid moiety in the skin, making it more permeable to the applied nutrient.
Applications & Uses
- Markets
- Applications
- Application Format
- Hair Care Applications
Properties
- Physical Form
- Odor
- Characteristic odor
- Appearance
- Off white low melting solid
- Insoluble in
- Water
- Soluble in
- chloroform, Methanol
- Chemical Properties
- Physical Properties
- Other Properties
- Microbiological Values
| Value | Units | Test Method / Conditions | |
| Tetrahydropiperine Content (HPLC - on dry basis) | 98.5 - 102.0 | % w/w | SLL/STP-T-001 |
| Impurities | max. 1.0 | % w/w | SLL/STP-T-001, By HPLC |
| Value | Units | Test Method / Conditions | |
| Loss on Drying (Dried at Room Temperature, Under Vacuum) | max. 1.0 | % w/w | USP <731> |
| Melting Range | 44.0 - 49.0 | °C | USP <741> |
| Value | Units | Test Method / Conditions | |
| Arsenic Content | max. 1 | ppm (μg/g) | USP <2232> |
| Cadmium Content | max. 1 | ppm (μg/g) | USP<2232> |
| Lead Content | max. 3 | ppm (μg/g) | USP<2232> |
| Mercury Content | max. 0.1 | ppm (μg/g) | USP<2232> |
| Value | Units | Test Method / Conditions | |
| Coliforms Count | max. 10 | cfu/g | BAM 2001, 8th Edition, Chapter 4 |
| Escherichia Coli | Negative | per 10g | USP <62> |
| Pseudomonas Aeruginosa | Negative | per 10g | USP <62> |
| Salmonella | Negative | per 10g | USP <62> |
| Staphylococcus Aureus | Negative | per 10g | USP <62> |
| Total Aerobic Microbial Count | max. 5000 | cfu/g | USP <61> |
| Total Yeasts and Molds Count | max. 100 | cfu/g | USP <61> |
Regulatory & Compliance
- Certifications & Compliance
- Chemical Inventories
Technical Details & Test Data
- Study Details
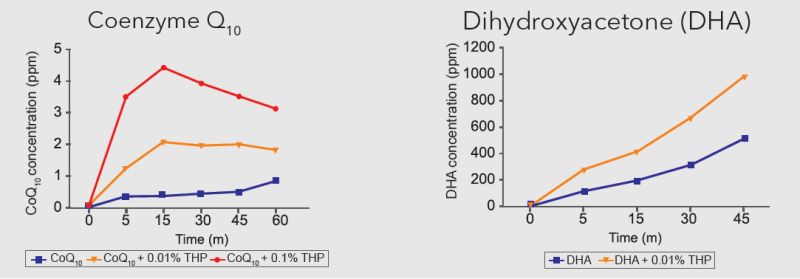
Cosmoperine® was found to increase the absorption of various actives including Tetrahydrocurcuminoids (30%), Forskolin (40%), Green tea polyphenols (30%), Betamethason (100%), Coenzyme Q10 (CoQ10) and Dihydroxyacetone (DHA) in an in vitro Franz cell diffusion assay.
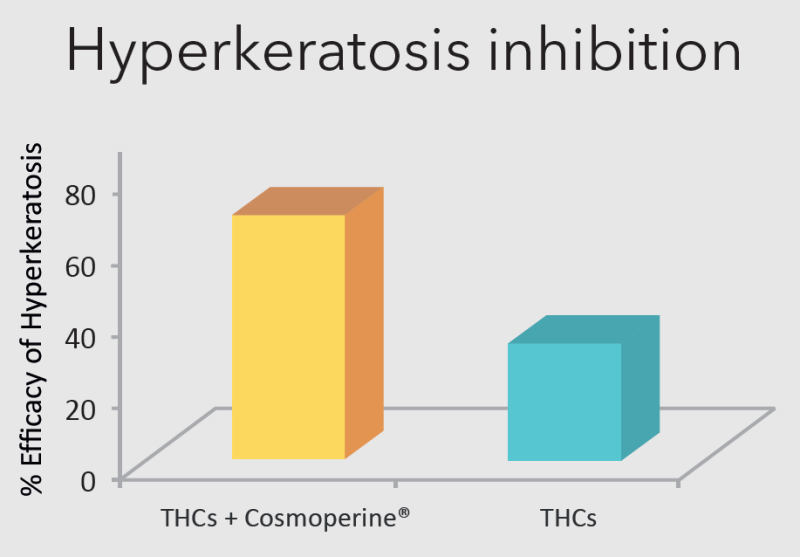
In a double-blind, randomized, non-cross over comparative clinical study, 20 healthy volunteers applied the creams containing Tetrahydrocurcuminoids (THCs)+Cosmoperine® and THCs alone for 3 months, found THCs+Cosmoperine® helped in the management of photo-damage to skin significantly better than THCs alone.
Packaging & Availability
- Regional Availability
Storage & Handling
- Shelf Life
- 5 Years
- Storage Condition
- Store between 10°C - 20°C (depending upon the destination)

