Knowde Enhanced TDS
Identification & Functionality
- Chemical Name
- INCI Name
- Cleaning Ingredients Functions
- Cosmetic Ingredients Functions
- CAS No.
- 1884575-91-0
- EC No.
- 813-944-0
- Technologies
Features & Benefits
- Benefit Claims
- Labeling Claims
- HII Features
- Product Benefits
- Broad spectrum activity
- Near zero probability of bacterial resistance
- Biocompatibility
- Low environmental concern
- Biodegradable
Applications & Uses
- Markets
- Applications
- Hair Care Applications
- Skin Care Applications
- Use Level
- 0.05-0.15%
- Application pH Range
- 3.5-10
Properties
- Appearance
- White to off-white powder
- Slightly Soluble In
- water
Technical Details & Test Data
- Impurities
Organic Impurities Chlorhexidine oxazinone analog 0.2% Specified unidentified impurity 1 0.2% Chlorhexidine amine 0.3% Chlorhexidine amine 1% Chlorhexidine urea 0.2% - Efficacy Tests
Comparison with Other Anti-Microbials
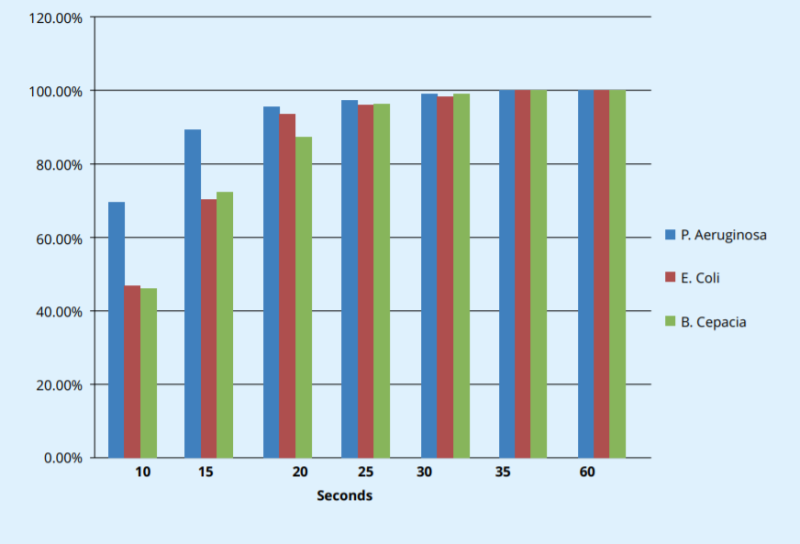
Test Organism SALIBACT Nanosilver Triclosan Triclocarban Effective against gram (+) bacteria ✓ ✓ ✓ ✓ Effective against gram (-) bacteria ✓ ✓ ✓ - Antifungal Properties ✓ ✓ ✓ - Fast acting (Time to complete kill) ✓ ✓ ✓ Biocompatibility (inert towards mammalian cells) ✓ - - - Multi-faceted mode of kill ✓ - - - Very low probability of bacterial resistance ✓ - - - Minimal environmental concerns ✓ - - - Controllable release (tunable solubility) ✓ - - - Cost Effective ✓ ✓ ✓ Challenge Test Result on Soap Bar using Salibact @ 0.1%
Safety & Health
- Toxicological Information
Acute and Genetic Toxicology Studies
- Acute Dermal Toxicology Studies – LD 50 greater than 2000mg/kg body weight
- Acute Oral Toxicity – LD50 greater than 2000 mg/kg body weight
- Salibact did not induce Gene Mutation
- Eye Irritation studies – Salibact was found to be a mild irritant.
- Acute Dermal Irritation studies – Salibact is a non-irritant

