Knowde Enhanced TDS
Identification & Functionality
- Ingredient Name
- Food Ingredients Functions
- Pharma & Nutraceuticals Functions
- CAS No.
- 25249-54-1
- Molecular formula
- (C6H9NO)n
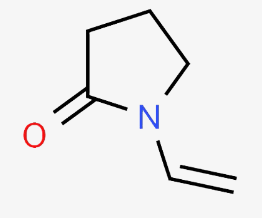
- Ingredients
- Polyvinylpyrrolidone
- Food Additive Number
- E 1201, INS 1201
- Product Families
- Chemical Structure
Features & Benefits
- Features
The water absorption rate increases with the increase of relative humidity. The formation of gel inhibits the penetration of water into the tablet, and the disintegration rate of the tablet slows down. Contrary to some other disintegrants, PVPP has no tendency to form gel at all because of its insolubility.
-
Complexation and solubilization
PVPP can form complexes with a variety of medicines and other substances. The formation process is reversible and will not form in alkaline media. It is easier to form complexes with those aromatic compounds containing phenyl and/or carboxyl groups. Therefore, in the pharmaceutical industry, it can improve the solubility and bioavailability of medicines, improve the taste of paracetamol, and absorb from tinctures and herbs. Removal of polyphenols and tannins, has the effect of resisting diarrhea, gastritis, ulcers, esophageal hiatus cancer and so on.
- Swelling and disintegration
PVPP has high capillary activity, so it can quickly absorb water into the tablet. Because the internal pressure (swelling pressure) exceeds the strength of the tablet, the tablet disintegrates instantly. And because there are folded molecular chains between the crosslinks, when water or aqueous solution penetrates, they are stretched by sudden impact and forced to separate immediately. As a result of swelling, the net volume increases (approximately doubled in 5 minutes), so that tablets prepared with PVPP tablet disintegrants disintegrate immediately.
-
Compressibility and adhesion
PVPP has excellent fluidity and plastic deformation, so they have excellent adhesion. This property gives good compressibility in spraying, drying, and wet granulation. Adding PVPP tablet disintegrant to the tablet, compared with commonly used starch, cellulose preparations and pectin compounds, the hardness and abrasion resistance of the tablet are not affected, and the capping of the tablet is not reduced. PVPP can also be effectively used as a tablet disintegrant in direct compression, without the use of lubricants or the addition of binders.
-
Capillary activity and hydration capacity
Research work has shown that PVPP as a tablet disintegrant has high capillary activity and hydration capacity, because of their large specific surface area (average 1.25 m2/g) and significant water absorption (high and rapid water absorption), Up to 60% or more).
-
Good compatibility
PVPP and a variety of organic and inorganic pharmaceutical excipients and active ingredients have very good compatibility. Solid drugs containing PVPP tablet disintegrants can use common tablet production techniques, including direct compression, It is easy to produce by spraying, drying and wet granulation.
-
Applications & Uses
- Applications
- Manufacturing Technology
- Food & Nutrition Applications
- Applications
Food
It can be used as a stabilizer for beer, liquor, wine, fruit juice and other beverages to extend its storage period.
Medicine
It is mainly used as a disintegrant for tablets, but also as a disintegrant and filler for pills, granules and hard capsules.
- Applications
PVPP-F can be used once, not renewable
Properties
- Appearance
- White or Nearly White
- Odor
- Odorless or Slightly Odor
- Insoluble in
- Water, Ethanol, Chloroform, Ether, Strong Alkali, Strong Acid
- Typical Properties
- Properties
Hygroscopic and easy to flow powder. It has very light swelling and excellent Physiological inertia, biocompatibility and good complexing ability with many kinds of substances.
| Value | Units | Test Method / Conditions | |
| Arsenic Content | max. 3 | — | — |
| Ash (Sulfate) | max. 0.2 | % | — |
| Boiling Point (760 mmHg) | 217.6 | °C | — |
| Copper + Zinc Content | max. 50 | — | — |
| Incomplete Order | max. 0.1 | % | — |
| Lead Content | max. 10 | — | — |
| Moisture Content | max. 6 | % | — |
| Nitrogen Content | 11.0-12.8 | % | — |
| PH value (1% aqueous solution) | 2021-05-11 | — | — |
| Soluble matter | max. 1.5 | % | — |
| Zinc Content | max. 25 | — | — |
Regulatory & Compliance
- Certifications & Compliance
- Enterprise Standard
Q/PGM002-2007 standard
Safety & Health
- Safety
Avoid contact with skin and eyes, do not inhale dust, do not ingest. If swallowed accidentally, seek medical assistance immediately to reduce dust generation and accumulation as much as possible, and ensure adequate ventilation.
Packaging & Availability
- Packaging Type
- Packaging
Packed in cardboard drum, net weight 20 kg/drum
Storage & Handling
- Storage
The product should be sealed and stored in a clean and dry warehouse, and attention should be paid to fire, water, sun, dust and pollution, etc.