Knowde Enhanced TDS
Identification & Functionality
- Chemical Name
- CASE Ingredients Functions
- Name
- 2,4,5,7-tetraiodo-3-hydroxy-6-fluorone
- CAS No.
- 142189-38-6
- Technologies
- Product Families
Features & Benefits
- Labeling Claims
- CASE Ingredients Features
- Advantages
- High absorptivity, low concentrations are needed (0.01-0.15% by weight)
- Cationic cured epoxides are not susceptible to oxygen inhibition and exhibit a tack free surface upon cure
- Capable of significant depth of cure > 1 inch
- Cure through UV opaque or pigmented substrates (e.g. Kapton)
- Initiator bleaching (bright red to pale pink/no color) upon light interaction can act as cure indicator
Applications & Uses
- Applications
- Cure Method
- Adhesive & Sealant Type
- Coating Type
- Ink & Toner Type
- Examples of H-Nu 535 uses
SU-8 photoresist
- Y. Lin, P.R. Hermann, and K.Darmawikarta, Appl. Phys. Lett. 86, 7, 071117 (2005)
- J.H. Moon, S.-M. Yang, D.J. Pine, and W.-S. Chang, Appl. Phys. Lett. 85, 18, 4184 (2004)
- D. Rodriguez Ponce, K Lozano, et al. J. Polym. Sci.: Part B: Polym. Phys. 48, 1, 47 (2010)
Properties
- Appearance
- Red Powder
- Typical Properties
| Value | Units | Test Method / Conditions | |
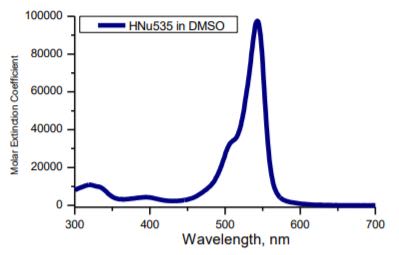
| Absorbance | max. 535 | nm | UV Visible spectroscopy |
| Melting Point | min. 270 | °C | Melting point Analysis |
| Molar Extinction Coefficient (535) | 91200 | nm | UV Visible spectroscopy |
Technical Details & Test Data
- Wavelength
- Applications Recommendations
- Direct solubility of H-Nu 535 in resins can be difficult, pre-dissolution in one of the following resins/solvents before adding resin is recommended:
Free-Radical Diluents Cationic Diluents DMAA VMOX THFA HDDA HPMA IBOA Heloxy61 Heloxy62 EOXA1 ECC2 Eponex1510 S S,T0 I I I I I I I P,T0 S,T0 1% of H-Nu 535 in diluent is: S-soluble Tº-requires heat (60ºC for several hours) P-partially soluble I-insoluble
1 = 3-ethyl-3-hydroxymethyloxetane
2 = 3,4-epoxycyclohexylmethyl-3,4
Note: DMAA is an excellent solvent for H-Nu 535, but it cannot be used in cationic polymerization as it inhibits process.
- The presence of any undissolved red particles is an indication of incomplete solubility. More heating (60℃) or stirring may help with incorporation, or pre-dissolution using different diluents is recommended.
Polymerization mechanisms
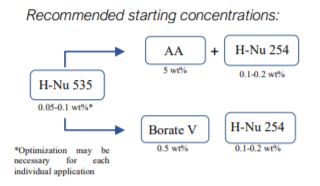
Free-Radical Cure (Acrylates)- H-Nu 535 concentration range spans from 0.05 to 0.2 wt.%, depending on a cure thickness. 0.05 wt% is recommended starting point for thick cure (>1mm) and 0.1wt% for thin cure.
- Amine acrylates (AA) are needed as co-initiators at 5-10 wt.%. If amine use is counterindicated (acidic adhesion promoter, outgassing), as an alternative, another co-initiator, Borate V (0.5 wt.%) may provide similar cure response as typical amine co-initiators
- H-Nu 254 iodonium salt is recommended for acceleration.
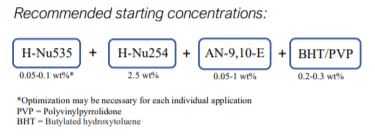
Cationic Cure (Epoxides)
- H-Nu 535 concentration range spans from 0.05 to 0.2 wt.%, with a good starting point at 0.1 wt.% based on solids.
- H-Nu 254 iodonium salt is needed to achieve cure in epoxide resins (sulfonium salts will not work).
- Accelerator AN-9,10-E can greatly enhance cure speed and sensitivity and is needed to achieve cure.
- Amines or DMAA solvent cannot be used due to the superacid “poisoning” effect.
- Caution & experimentation is required at higher temperatures as it can lead to thermal ground state polymerization. Additional stabilizers, depending on the photopolymerization matrix can be used. As an example, 0.2 wt.% of PVP+BHT* mixture (1:1) can be added to glycidyl epoxide and 0.3 wt.% of PVP+BHT mixture (1:1) to cycloaliphatic epoxide.
- Structure