Knowde Enhanced TDS
Identification & Functionality
- Chemical Family
- Enzyme Type
- Ingredient Name
- Food Ingredients Functions
- Ingredients
- Protease, Wheat Flour
- Food Additive Number
- E 1100, INS 1100
- Technologies
- Product Families
- Origin
Non-pathogenic, non-gene-modified Bacillus subtilis strain (GRAS Status)
Features & Benefits
- Labeling Claims
- Food Ingredients Features
- Properties
- Gluten weakening
- Reduces energy input during kneading
- Reduces resting time
- Imparts better chewing characteristics
- Reduces hairline cracks
- Enhances structure and browning
Applications & Uses
- Markets
- Applications
- Applications
- Biscuit and cracker production
- Dosage
6 - 15 g on 100 kg flour. Particular processes may require as much as 24 g or as little as 3 g. The optimum dosage has to be established by baking trials (see also page 2, bottom).
Properties
- Appearance
- Powder
- Odor
- Typical slightly fermented
- Chemical Properties
- Typical Properties
- Microbiological Values
- Nutritional Information
- Nutritional Data
Description Value Unit/100g Energy 1.485 kJ Energy 350 kcal Fat 1 g of which: saturated fatty acids (FA) < 0.2 g Carbohydrates 69 g of which: Sugar 0.5 g Fiber 3.6 g Protein (N x 6,25) 14.5 g Salt (Sodium x 2.5) 0.011 g Alcohol 0 g Trans fatty acids 0 g Sodium < 0,005 g
| Value | Units | Test Method / Conditions | |
| Arsenic Content | max. 3 | mg/kg | AAS |
| Cadmium Content | max. 0.5 | mg/kg | AAS |
| Lead Content | max. 5 | mg/kg | AAS |
| Loss on Drying | max. 14 | g/100g | AAS |
| Mercury Content | max. 0.5 | mg/kg | Method |
| Value | Units | Test Method / Conditions | |
| Protease | 420 - 520 | u/g | Casein hydrolysis pH 8 |
| Value | Units | Test Method / Conditions | |
| Escherichia coli | Negative | cfu/g | M. Colif.-Agar 36°C/48h |
| Salmonella | Negative | cfu/25g | DIN EN ISO 6579 |
| Total Plate Count | max. 50000 | cfu/g | DIN EN ISO 4833 |
| Value | Units | Test Method / Conditions | |
| Energy | 1.485 | kJ/100 g | — |
| Fat Content | 1 | g/100g | — |
| Saturated Fatty Acids | max. 0.2 | g/100g | — |
| Carbohydrates | 69 | g/100g | — |
| Sugar | 0.5 | g/100g | — |
| Fibre | 3.6 | g/100g | — |
| Protein (N x 6.25) | 14.5 | g/100g | — |
| Salt (Sodium x 2.5) | 0.011 | g/100g | — |
| Alcohol | 0 | g/100g | — |
| Trans Fatty Acids | 0 | g/100g | — |
| Sodium Content | max. 0.005 | g/100g | — |
Regulatory & Compliance
- Regulatory Information
Allergen Information: contains the allergen gluten according to Regulation 1169/2011/EG, Annex II
Health Information: The products are suitable for human consumption if used accordingly to recommendations and officially allowed to be sold within Germany and the EU. The product is food grade and conforms with the current european food law.
GMO: This product is not a genetically modified organism (GMO), nor does it contain any GMO or any recombinant DNA. All the non-enzymatic constituents of the product (e.g. carriers, stabilizing agents, free-flowing agents etc.) originate from non-genetically modified sources. Due to the ubiquitous presence of GMO organisms, absence of minor traces of GMO material cannot be guaranteed, but in any case, the content of unintentionally present GMO material will be below 0.9 %, in accordance to EU guidelines 1830/2003 and 1829/2003. The product is used as processing aid in food manufacturing processes, and therefore labeling is not required in the EU. National regulations must be observed.
Product suitability:
Yes No Vegetarian X Vegan X BSE/TSE Information: The mentioned product does not represent a BSE (Bovine Spongiform Encephalopathy) and/or TSE (Transmissible Spongiform Encephalopathy) risk.
Irradiation: Neither the product nor its ingredients have been treated with ionizing radiation according to Directive 1999/2/EC and 1999/3/EC.
Nanoparticles: The product does not contain ingredients in the form of engineered nanomaterials as defined in Regulation (EU) 2015/2283.
Contamination/Residues: The product complies with: Regulation (EC) No 396/2005 and its amendments regarding maximum levels for pesticide residues in foodstuffs. Regulation (EC) No 1881/2006 and its amendments regarding especially maximum levels for heavy metals, mycotoxins, dioxins and PCB in foodstuffs.
Conformity of the Packaging: The packaging material complies with the provisions of Regulation (EC) No 1935/2004 and Regulation (EU) No 10/2011.
Technical Details & Test Data
- Activity
Bacterial protease 500 u/g (casein hydrolysis, pH 8.0) pH optimum 6 – 8 (see Fig. 2 and 3) Temperature optimum 40 – 60°C (see Fig. 4 and 5) Comparison of cookies baked w/ and w/o Sternzym BK 18222 SF

Sternzym BK 18222 SF - Neutral bacterial protease from B. subtilis
Temperature/pH activity and stability dataFig. 2: pH-activity curve for STERNZYM BK 18222 SF
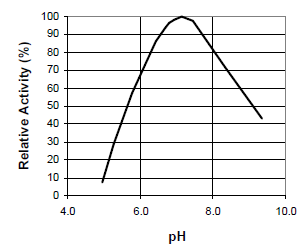
Fig. 3: pH-stability curve for STERNZYM BK 18222 SF
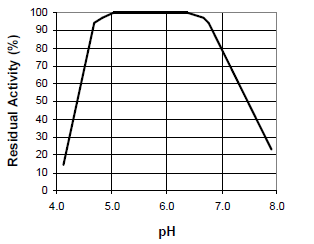
Fig. 4: Temperature-activity curve for 120 STERNZYM BK 18222 SF
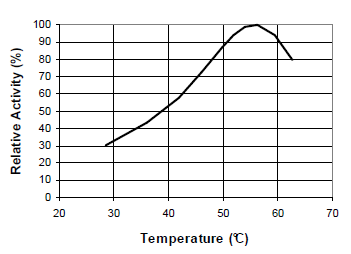
Fig. 5: Temperature-stability curve for STERNZYM BK 18222 SF
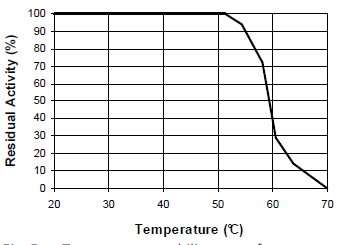
Optimization
The exact dosage has to be established in production trials. We recommend to start with a low dosage of the enzyme, approx. 3g per 100 kg, and to increase the addition in iterative steps of 1 or 2g until the desired effect is obtained. As the enzyme activity improves with increasing water availability and temperature, minor modifications of the process should be considered if the desired effects cannot be achieved.
Packaging & Availability
- Packaging
Cardboard box or multiply paper bag with PE liner, 20 kg net, 25 kg net.
Storage & Handling
- Storage & Shelf Life
Min. 18 months if stored cool and dry in closed original packing, activity loss on storage less than 10% per year