Knowde Enhanced TDS
Identification & Functionality
- INCI Name
- Cosmetic Ingredients Functions
- Encapsulated Material
- Pure Retinol
- Technologies
- Product Families
- Auxiliaries
Magnesium stearate (and) silica silylate (and) silica
Features & Benefits
- Benefit Claims
- Labeling Claims
- Product Attributes
Why You Should Use CelluCap™ RL in Your Formulas?
- Increases retinol’s stability and potency
- Reduces the presence and size of dark spots
- Less irritating when compared to free retinol (CIT tested at final free retinol level of 0.1%)
- Facilitates easy formulation processes
- Improves skin homogeneity
- Product Background
Innovating Retinol Usage in Skincare Formulas
Pure Retinol is one of the golden standards in skincare when it comes to anti-aging formulas. Cosmetic manufacturers throughout the world rely on its unique properties when creating their skincare products geared at reducing hyperpigmentation, revitalizing skin, reducing wrinkles, and creating a younger-looking complexion. However, Retinol’s sensitivity to light, oxygen, and heat makes it very unstable for formulators to use and its instability can decrease its efficacy when applied.
CelluCap™ RL uses Tagra’s patented microencapsulation technology to keep Retinol separate from the environment, with the microcapsules only breaking upon application on the user’s skin. In this way, Tagra’s technology is able to retain Retinol’s efficacy throughout the production process and the product's shelf life, keeping it active until use.
Applications & Uses
- Markets
- Applications
- Application Format
- Skin Care Applications
- Use Level
- 1 - 3%
- Formulating with CelluCap™ RL
For best practice in any cosmetic formulation, CelluCap™ RL should be gradually added at the last stage of formulation preparation, using a simple paddle/anchor type mixer at 70- 300rpm. Avoid using high shear mixing (homogenizer) and temperature exceeding 50°C. No nitrogen blanket is needed while formulating with CelluCap™ RL.
Properties
- Physical Form
- Appearance
- Free flowing powder
- Typical Properties
| Value | Units | Test Method / Conditions | |
| NOI | 0.52 | — | — |
| Payload (Pure Retinol) | 9 | % | — |
Regulatory & Compliance
- Certifications & Compliance
Technical Details & Test Data
- The Powerful Effect of CelluCap™ RL
CelluCap™ RL was tested clinically over 8 weeks on 22 patients. Tested formulation contained a final retinol concentration of 0.1%. The results below show a significant decrease in the number and area of hyperpigmented spots by 18.3% and 18.5%, respectively after 56 days treatment (D56), compared to baseline values before treatment started (D0). Likewise, skin homogeneity increased by 21.5%
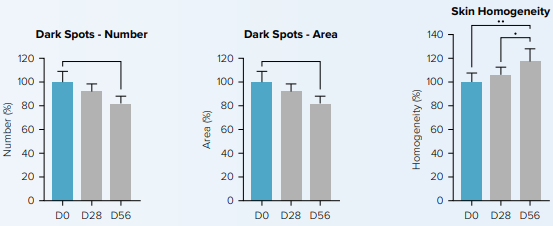
Dark spot areas were also measured using skin pigmentation analysis as depicted below.

Pigmentation

Pigmentation Analysis
Storage & Handling
- Storage Conditions
Closed container below 4°C.

