Knowde Enhanced TDS
Identification & Functionality
- Chemical Family
- Chemical Name
- Base Chemicals Functions
- CASE Ingredients Functions
- CAS No.
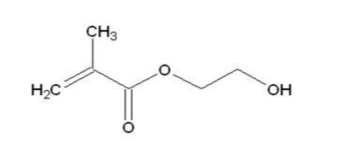
- 868-77-9
- EC No.
- 212-782-2
- Technologies
- Product Families
- Chemical structure
Applications & Uses
- Applications
- Applications
Bisomer® CL HEMA is widely used in the preparation of long-term wear and disposable contact lenses, intra-ocular lenses and diverse biomedical applications. Bisomer® CL HEMA is an ideal starting point in the formulation of biomedical hydrogels. Our organization offers dedicated monomer purification units for the production of high purity monomers in an environment controlled for air purity. We also offer customized products for biomedical and contact lens applications and large batch sizes which reduce customer testing requirements and provide greater batch to batch consistency. GEO Specialty Chemicals has monomer expertise from over 40 years of experience as a world leading supplier of hydroxyl monomers with ISO 9001:2000 and ISO 14001 accreditation.
Properties
- Appearance
- Clear liquid
- Typical Properties
| Value | Units | Test Method / Conditions | |
| Water content (mass) | max. 0.2 | % | (E203; 23-01) |
| Inhibitor (MEHQ) content (mass) | 15 - 30 | ppm | (D3125; 13-08) |
| Diester EGDMA % (mass) | max. 0.14 | % | (08-09) |
| Diethylene glycol methacrylate (mass) | max. 1.0 | % | (08-09) |
| Assay (mass) | 99.0 min | % | (GC; 08-09) |
| Color | max. 10 | Pt/Co | (D1209; 03-18) |
| Methacrylic acid (mass) | max. 0.05 | % | (E202; 01-01) |
| Molecular weight (av) | 130 | g / mol | — |
Regulatory & Compliance
- Certifications & Compliance
- Chemical Inventories
- Quality Standards
- Registration & Regulatory Information
Regulatory Status
AICS yes
DSL yes
ENCS yes
EU REACH Registration No. 01-2119490169-29-0002
IECSC yes
KECI yes
PICCS yes
TSCA yes
Food Packaging Approvals
EU 10/2011 Listed
FDA CFR 21 § 175.105, 177.1010
FDA CFR 21 § 175.300, 176.180 (Listed as a component of a polymer)
BfR Recommendations XIV, XXII, XXXVI
Swiss Ordinance 817.023.21 – Listed (Part A)

