Knowde Enhanced TDS
Identification & Functionality
- Active Component
- INCI Name
- Cosmetic Ingredients Functions
- Pharma & Nutraceuticals Functions
- CAS No.
- 331-39-5
- Molecular formula
- C9H8O4
- EC No.
- 206-361-2
- Product Families
- The mechanism of brightening function of caffeic acid is primarily based on below two facts
- The chemical structure of caffeic acid is very similar to L-tyrosine. Therefore, caffeic acid can be degraded by tyrosinase to become a competitive inhibitor against melanin.
- Caffeic acid can inhibit melanin production by inhibiting the signal path between tyrosinase and MITF (microphthalmia-associated transcription factor).
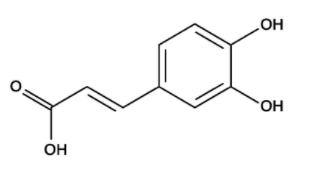
- Chemical Structure
Features & Benefits
- Benefit Claims
- Benefit Claims (Health)
- Labeling Claims
- Why choose Viablife Caffeic Acid
- The purity of most caffeic acid available on market is between 70%-90%.
- The color is dark yellow which is considered caused by impurities inside.
- So it's very difficult to apply this kind of quality caffeic acid in cosmetic product.
- Viablife uses industry-leading Biosynthesis and Fermentation technology to obtain high purity (>99%) caffeic acid.
- The color of our caffeic acid is light yellow which only has very little effect on the color of cosmetic products applied in.
Applications & Uses
- Applications
- Recommended use in cosmetic
- Rinse off : <0.4%
- Leave on: <0.25%
Properties
- Appearance
- Light Yellowish Crystallization
- Insoluble in
- Water
- Soluble in
- Ethanol
- Typical Properties
- Specifications
| Value | Units | Test Method / Conditions | |
| Assay | min. 99.0 | % | — |
| Heavy Metals (as Pb) | max. 20 | ppm | — |
| Loss on Drying | min. 1.0 | % | — |
| pH of Solution (109% water solution) | 5.5 - 7.5 | — | — |
| Purity | min. 99 | % | HPLC, LC-MS |
| Value | Units | Test Method / Conditions | |
| Molecular Weight | 103.12 | — | — |
Technical Details & Test Data
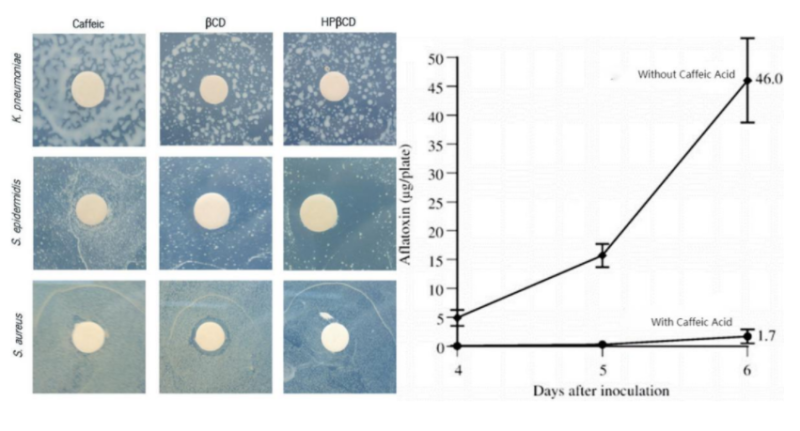
- Anti-Bacteria
Caffeic Acid shows great ability in inhibiting growth of microbe especially Aflatoxin.
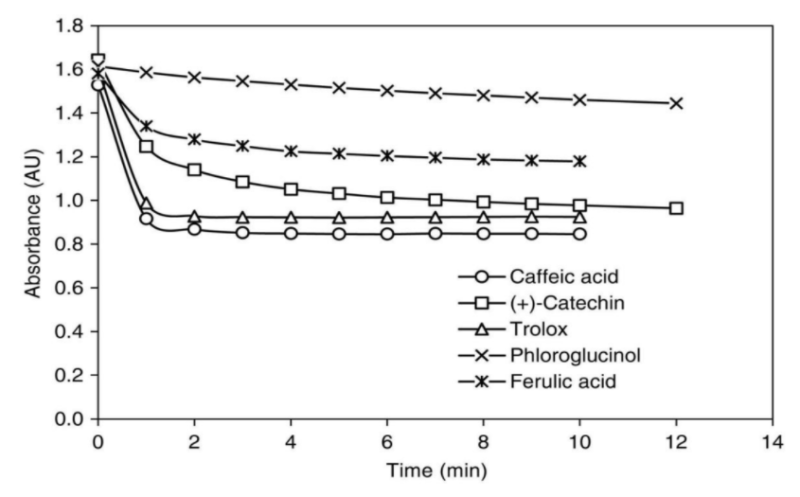
- Anti-Oxidant
Caffeic Acid has extraordinary capability of scavenging free radicals. In DPPH and ABTS experiments it prevails other phenolic acid compound and cinnamic acid derivatives.