Knowde Enhanced TDS
Identification & Functionality
- Active Component
- Ingredient Name
- Pharma & Nutraceuticals Functions
Features & Benefits
- Benefit Claims (Health)
- Labeling Claims
- Food Ingredients Features
- Product Highlights
- 100,000X aqueous solubility
- 3.7X oral bioavailability
- Scientifically proven efficacy
- Unique absorption mechanism
- 2023 US FDA New Dietary Ingredient
- Proven Efficacy
Applications & Uses
- Applications
- Food & Nutrition Applications
- Use Level
- 40 - 80 mg
Properties
- Physical Form
- Solubility
- Appearance
- White to pale yellow fine powder
- Chemical Properties
- Physical Properties
- Microbiological Values
- Heavy Metals
| Value | Units | Test Method / Conditions | |
| Water-soluble Isoflavone Derivatives | min. 70 | % | QA_Q02_AH1,HPLC |
| Arsenic Content | max. 2 | ppm | MOHWH0014.03 |
| Value | Units | Test Method / Conditions | |
| Loss on Drying | max. 6 | % | QA-Q01-AH1 |
| Value | Units | Test Method / Conditions | |
| Yeast & Molds Count | max. 102 | cfu/g | MOHWM0008.01 |
| Total Aerobic Plate Count | max. 103 | cfu/g | MOHWM0014.01 |
| Salmonella | Negative | /25g | MOHWM0025.01 |
| Escherichia Coli | Negative | /g | MOHWM0023.02 |
| Enterobacteria | max. 10 | cfu/g | MOHWM0028.00 |
| Value | Units | Test Method / Conditions | |
| Lead Content | max. 20 | ppm | MOHWH0014.03 |
Regulatory & Compliance
- Certifications & Compliance
Technical Details & Test Data
- Pharmacokinetic Profile
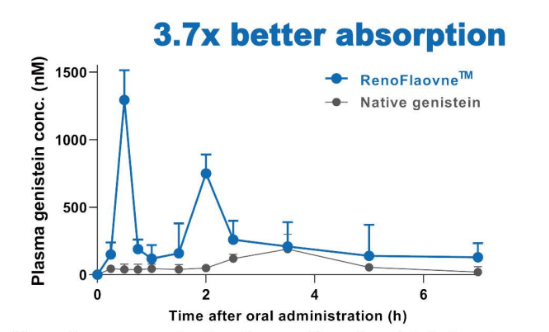
Mean plasma concentration-time profiles of genistein in rats after oral adminstration of native genistein and RenoFlavone™ at 3.7 u mol/kg bw. Data are mean SE (n = 4).- Animal Study Improve Menopausal Bone Health
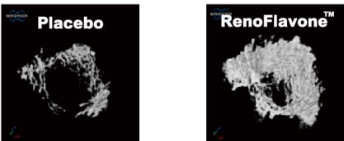
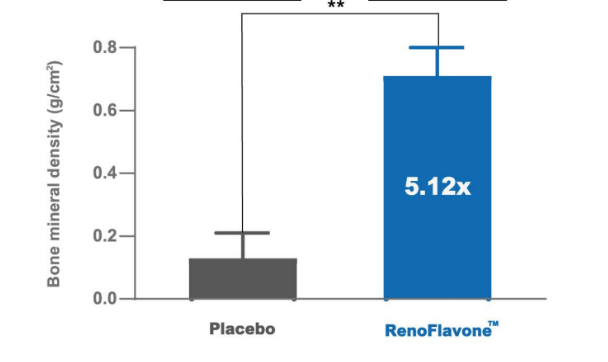
Osteoprotective effect of RenoFlavone™, a derivative of genistein with high bioavailability, in ovariectomized rats. Mi ovarietomized rats. Bet of plate and entral met wip (15.2 umol/kg bw/day) after 12-week treatment. Data are mean ‡SD, n = 6. Value are mean‡SEM. ** p < 0.01. RenoFlavone™ vs placebo group (paired t-test).- Human study Relieve menstrual discomfort
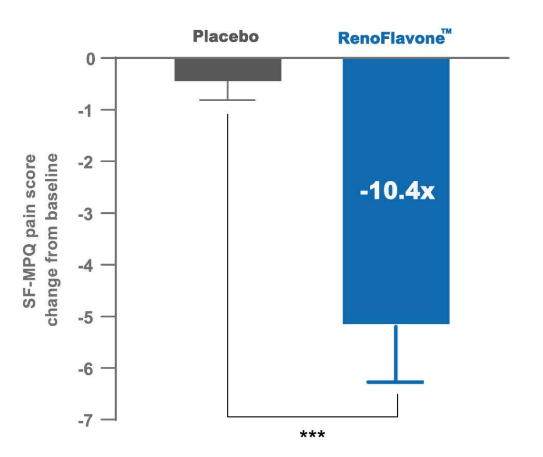
A randomized controlled trial of RenoFlavone™ in the treatment of primary dysmenorrhea. Effects of RenoFlavone™ (20 mg/ day) and placebo on menstrual pain score (Short form McGill pain questionnaire score, SF-MPQ) in first month (The trial is ongoing). Value are mean SEM, n = 40. Comparison of changes between the study groups (chi-square test). Significance of double star symbol is *** p< 0.001.
Packaging & Availability
- Packaging Type
Storage & Handling
- Shelf Life
- 3 years
- Storage and Shelf Life Conditions
Store in aluminum bag at controlled room temperature for 3 years.
