Knowde Enhanced TDS
Identification & Functionality
- Chemical Name
- Technologies
Features & Benefits
- Labeling Claims
Applications & Uses
- Markets
- Applicable Processes
Properties
- Physico-Chemical Properties
- Note
* After activation at 550°C for 3 hours
| Value | Units | Test Method / Conditions | |
| Al₂O₃ | 72.0 | % | — |
| Na₂O | 0.002 | % | — |
| Loose Bulk Density | 670 - 750 | g/l | — |
| Packed Bulk Density | 800 - 1100 | g/l | — |
| Particle Size (d50) | 60.0 | μm | — |
| Surface Area * | 230.0 | m²/g | Brunauer-Emmett-Teller Method |
| Pore Volume* | 0.5 | ml/g | — |
| Crystallite Size (120) | 5.5 | nm | — |
Technical Details & Test Data
- Processing of CATAPAL Products
The unique ability to adjust the physical properties of the boehmites makes them perfect for a variety of different end use applications requiring different types of processing.
Extrusion Behavior:
- Extrusion is a key use for these types of alumina hydrates due to their ability to form excellent extruded supports. This application takes advantage of the ability to peptize in the presence of organic/inorganic acids.
- A wide variety of extrudate properties can be obtained by using different grades of CATAPAL products and by controlling the formulation and extrusion variables. Optimum formulations depend on the exact choice of mixer, extruder and powder properties. General extrusion guidelines can be recommended by our technical staff.
- Our unique ability to adjust the peptization behavior means that the gelling behavior of CATAPAL products can be modified to supply a product optimized for our customers’ own unique set of processing conditions. Nitric acid gelation time (NAG) is a valuable tool for selecting the right material for your processing conditions.
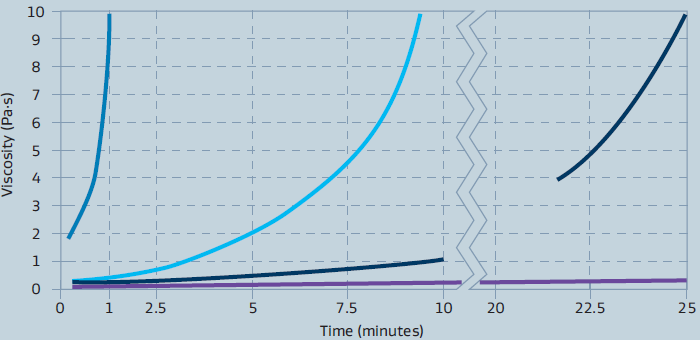
- NAG is the time required for a concentrated dispersion of boehmite to reach a certain viscosity by peptization. This behavior is shown in figure 6 and illustrates the range of NAGs we are able to produce on our customers’ request. The graph is not meant to indicate the only NAGs available.
Figure 6: Viscosity behavior of boehmite dispersions having different NAG values
- Advantages of CATAPAL Alumina Hydrates
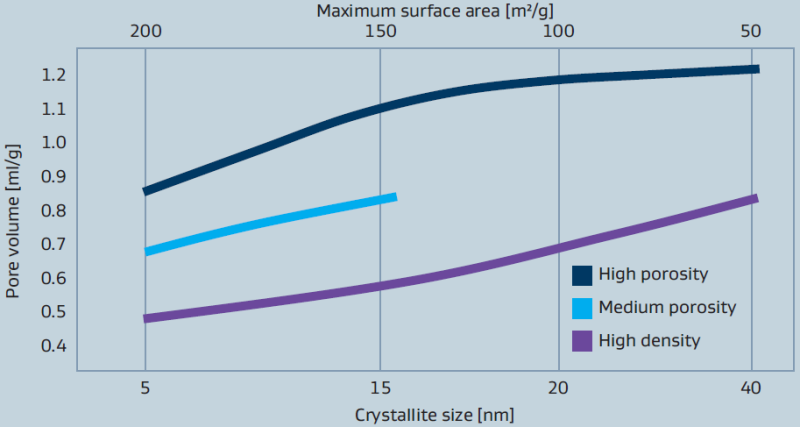
Sasol pioneered processes to convert aluminium metal to synthetic alumina hydrates of high-purity (figure 1). Unlike other alumina manufacturing processes that start with bauxite derivatives, our processes yield aluminas with significantly lower levels of common impurities such as iron, sodium and silica (as shown in table 1). Additionally, our 50 years of alumina manufacturing experience allows us to control and adjust physical properties such as surface area and porosity (figure 2), particle size (figure 3), crystal size and shape (figure 4) and peptization behavior (figure 5) so that our customers are not limited in their thinking for possible uses for the aluminas. Ultra-high-purity grades (PURAL UHPA) are available upon request.
Figure 2: Relationship between crystallite size, pore volume
Chemical purity of CATAPAL Alumina Hydrates
Impurity ppm (typical) Na2O <20 Fe2O3 50-100 SiO2 50-120
Figure 3: Examples of particle size distributions of CATAPAL
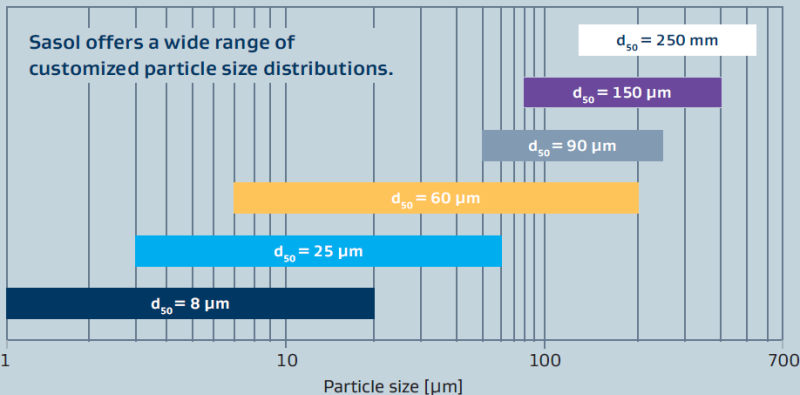
Figure 4: Boehmite of different crystal shapes
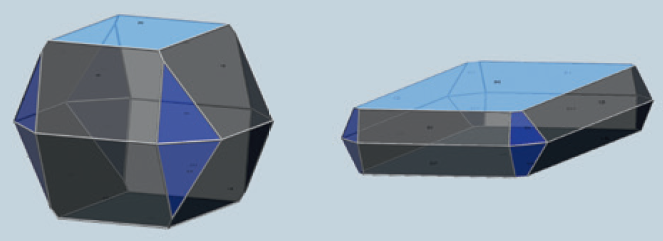
Figure 5: TEM images of boehmite with different crystallite sizes

- Alumina Production Process
Sasol Inorganics produces high- and ultra-high-purity aluminas primarily through synthetic aluminum alkoxide processing routes. The alumina is produced either as a co-product with synthetic linear alcohols (Ziegler method) or directly from aluminum metal (on-purpose route). Several production steps must be completed to produce the different alumina-based products. In the first step, an aqueous intermediate (alumina slurry) is produced, which is further tailored in the subsequent processing steps to obtain the various products sold on the market. These can be alumina hydrates, calcined aluminas, and doped versions thereof.
Figure 1: Schematic for the Alumina Manufacturing Process
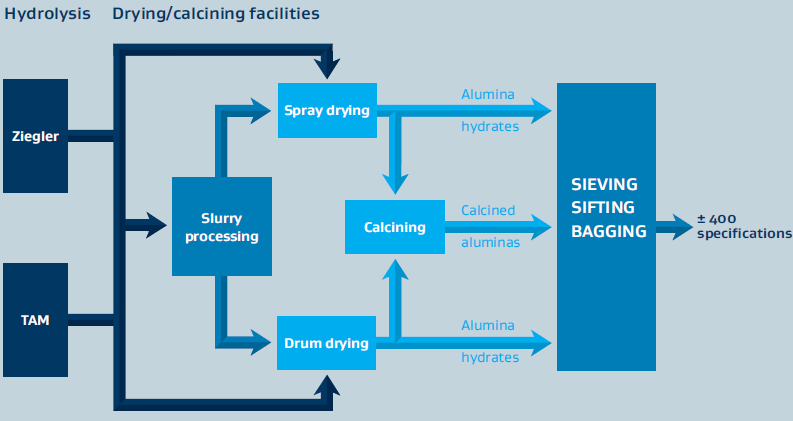
- Calcination
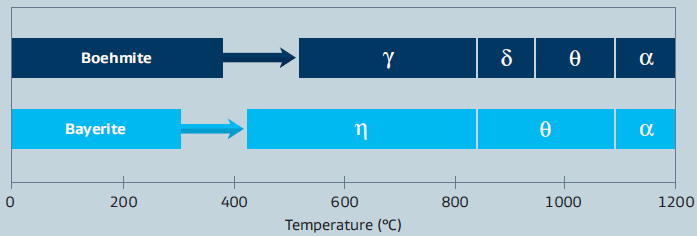
The final crystalline phase and physical properties of calcined aluminas depend on the initial crystalline properties of the starting alumina hydrate as well as the calcination temperature. The sequence of phase transitions of boehmite and bayerite are shown in figure 7.
Physical properties such as crystalline phase, surface area and porosity can be altered significantly by varying the calcining time and temperature. Applied temperatures are typically between 600°C and 1,000°C. This process results in the loss of physisorbed and crystal water.
Figure 7: Sequence of phase transitions
Storage & Handling
- Storage & Handling Information
Sasol pioneered processes to convert aluminum metal to synthetic alumina hydrates of high-purity (figure 1). Unlike other alumina manufacturing processes that start with bauxite derivatives, our processes yield aluminas with significantly lower levels of common impurities such as iron, sodium and silica (as shown in table 1). Additionally, our 50 years of alumina manufacturing experience allows us to control and adjust physical properties such as surface area and porosity (figure 2), particle size (figure 3), crystal size and shape (figure 4) and peptization behavior (figure 5) so that our customers are not limited in their thinking for possible uses for the aluminas. Ultra-high-purity grades (PURAL UHPA) are available upon request.
Figure 2: Relationship between crystallite size, pore volume

Chemical purity of CATAPAL Alumina Hydrates
Impurity ppm (typical) Na2O <20 Fe2O3 50-100 SiO2 50-120
Figure 3: Examples of particle size distributions of CATAPAL

Figure 4: Boehmite of different crystal shapes

Figure 5: TEM images of boehmite with different crystallite sizes


