Knowde Enhanced TDS
Identification & Functionality
- Carrier
- Chemical Family
- Cosmetic Ingredients Functions
- Technologies
Features & Benefits
- Benefit Claims
- Labeling Claims
- Product Background
Silasoma is aqueous dispersions (containing 60% microcapsule as active component) in microcapsules having an average particle diameter of 2 micrometers, which encapsulate UV absorbers. Encapsulating the UV absorbers provides a soft and light after-feel. It also allows for a surfactant-free and safer sun protection formulation.
Silasoma is water dispersion of microcapsules, encapsulated UV absorbers
Concern related to the safety of UV absorbers and their heavy oily feeling on the skin lead us to encapsulate the raw material, we have developed with our own original patented technology, resulting in the successful development of Silasoma, an ingredient that protects against UV rays and is gentle on the skin. In addition to being very safe, Silasoma also has a useful feature in that it provides a soft and light after-feel that is devoid of stickiness, and that it allows the formulation of surfactant-free sun protection products. It can also be incorporated into hair care products.
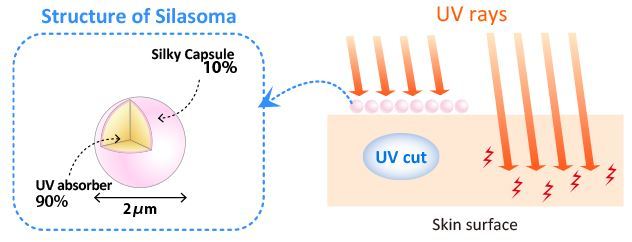
- Product Characteristics
- Organic UV absorber, which causes greasy and irritating to skin, is encapsulated by silk component.
- This protects sensitive skin from UV sufficiently with silky and smooth usability.
- Special cleansing does not be needed to be remove UV protection cosmetics containing Silasoma from skin.
Applications & Uses
- Markets
- Hair Care Applications
Properties
- Typical Properties
| Value | Units | Test Method / Conditions | |
| Pentylene Glycol Content | 3 | — | — |
| Tocopherols Content | max. 0.1 | — | — |
Technical Details & Test Data
- Efficacy Tests
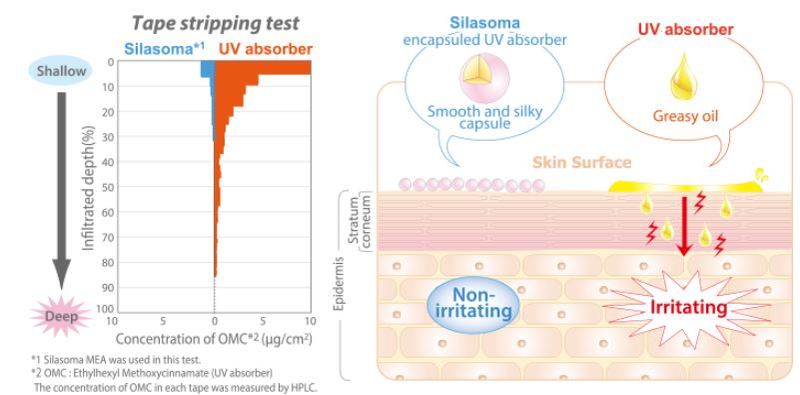
- We tested the safety by encapsulation of UV absorbers.
- A lotion containing Silasoma and another lotion, which was formulated with the UV absorber in the same amount as that inside the microcapsule, were applied to each arm, respectively. Both were left on the arm for 60 minutes. Afterwards, any formulation remaining on the skin was wiped off. The stratum corneum cells were stripped off with tape, and the amount of UV absorbers adhering to the stratum corneum was measured. The stripping with tape was repeated to examine the degree of penetration into the stratum corneum.
Packaging & Availability
- Regional Availability

