Knowde Enhanced TDS
Identification & Functionality
- Active Component
- Country of Origin
- Ingredient Name
- Pharma & Nutraceuticals Functions
Features & Benefits
- Food Ingredients Features
- Product Highlights
Ginseng is native to the mountains of Northeast China, the East Russia, and the Korean Peninsula. Since ancient times, ginseng has been known as the"King of Herbs". In Western countries, ginseng is also known as"the panacea"("Panax"means"treatment of all diseases"). It is a protected plant both in China and Russia now. Ginseng is a slow growing perennial, and the roots are usually harvested by hand when the plant is five/six years old. Panax Ginseng Extract is an extract or concentrated powder obtained by extracting and concentrating the roots or leaves of Panax ginseng C.A. Meyer. Panax Ginseng Extract's main active ingredient is ginsenoside, ginseng polysaccharide, sterol, amino acid, etc. In modern scientific research, the popular efficacy research on ginseng and Panax Ginseng Extract's active ingredients at home and abroad mainly focuses on relieving physical fatigue, delaying aging, weight management, blood pressure, anti-inflammatory, anti-tumor, anti-oxidation, and glucose and lipid metabolism. Nowadays, Panax Ginseng Extract has been widely used in medicines, health products, functional foods and cosmetics.
- Features
- 10 years production experience. Complete specifications.
- Patented technology for removing pesticide residues.
- Multiple product forms. One-stop solution service.
Applications & Uses
- Applications
- Food & Nutrition Applications
- Applications
- Granule
- Gummy
- Powder
- Freeze-dried Bites
- Tablet
- Functional Drink
- Hard Capsule
- Energy Drink
- Soft Capsule
- Effervescent Tablets
Properties
- Physical Form
- Appearance
- It should be off-white to brown yellow fine powder, with special odor and taste, without obvious visible impurities.
- Soluble in
- Water (in100mL, 1g)
- Typical Properties
- Microbiological Values
- Assay
- Heavy Metals
| Value | Units | Test Method / Conditions | |
| Loss On Drying | max. 5.0 | % | Ch.P<0831> |
| Ash Content | max. 5.0 | % | Ch.P<2302> |
| Particle Size (80 Mesh Sieve) | min. 95 | % | Ch.P<0982> |
| Native Extract Powder | 25.0 | % | — |
| Maltodextrin Content | 75.0 | % | — |
| Value | Units | Test Method / Conditions | |
| TABC | max. 1000 | cfu/g | Ch.P<1105> |
| TMYC | max. 100 | cfu/g | Ch.P<1105> |
| E. Coli | Absent | /10g | Ch.P<1106> |
| Salmonella | Absent | /10g | Ch.P<1106> |
| Value | Units | Test Method / Conditions | |
| Ginsenosides (Rg1+Re+Rb1+Rc+Rb2+Rf+Rd) | 1 - 60 | % | Ch.P<0512>/Ch.P<0401> |
| Value | Units | Test Method / Conditions | |
| Total Heavy Metals | max. 10.0 | ppm | Ch.P<0821> |
| Lead Content | max. 3.0 | ppm | Ch.P<2321> |
| Arsenic Content | max. 2.0 | ppm | Ch.P<2321> |
| Mercury Content | max. 0.1 | ppm | Ch.P<2321> |
| Cadmium Content | max. 1.0 | ppm | Ch.P<2321> |
Regulatory & Compliance
Technical Details & Test Data
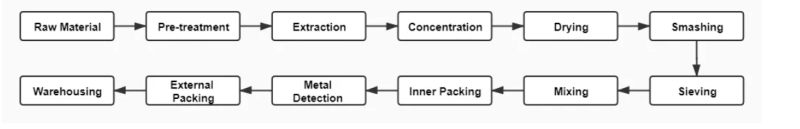
- Flow Chart