Knowde Enhanced TDS
Identification & Functionality
- Chemical Family
- Chemical Name
- Cleaning Ingredients Functions
- Pharma & Nutraceuticals Functions
- CAS No.
- 124-04-9
- EC No.
- 204-673-3
- Product Families
Features & Benefits
- HII Features
- Materials Features
Applications & Uses
- Applications
- Dosage Form
- Base Chemicals End Uses
- Home Care Applications
- Applications
- As a monomer in nylon, paper additives, copolyamides, terpolymers, and unsaturated polyester resins (UPRs)
- In polymer additives for epoxy curing agents and plasticizers.
- As a chemical intermediate in synthesis of polyesters/diesters, polyester polyols, adiponitrile, cyclopentanone, 1,6-hexanediol, and dimethyl sebacate.
- In other applications such as solvents, lubricants, electronics, soil conditioners, glass protection agents, briquetting agents, leather tanning agents, flue gas desulfurization scrubbers, and cleaning aids.
- Polyamides
Nylon, a class of aliphatic polyamides, is the single largest commercial application for Adi-pure® high purity adipic acid. Nylon 66 (N66), or polyhexamethyleneadipamide, and Nylon 6 (N6), or polycaprolactam, are the most economically important nylons. Both can be converted to fibers, used for carpets, apparel, tire cord, and industrial applications, or to engineering resins, used for automotive parts, electrical and electronic applications, machine parts, films, wire coatings, and monofilament. Fibers are produced as continuous filament yarn and as staple; resins are produced for use in injection molding or extrusion. Engineering resins can also be glass reinforced, mineral filled, or otherwise modified to improve performance.
The differences between N66 and N6 are often subtle, but, in general, N66 has higher tensile strength, greater hardness, higher heat deflection temperature, and a lower rate of water absorption. In fiber applications, nylon properties of value are low moisture absorption and abrasion resistance, and its overall properties are dependent on spinning speed, draw ratio, and the types of additives or finishes used. For engineering resins, nylon is particularly valuable because of its low coefficient of friction, excellent abrasion resistance, good chemical resistance, and high impact strength.
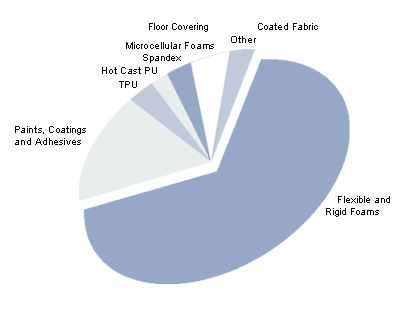
- Polyurethanes
Polyuethanes (PU´s) are one of the most versatile plastics available today, offering a wide range of properties suitable for many end-uses. This is due to the fact that they are segmented polymers which combine the physical properties of the hard and soft segments. PU´s are available in several forms, each with different potential applications.
The majority of PU´s are produced from the reaction of either a polyester or polyether polyol with a diisocyanate and chain extender to give a variety of physical and chemical properties. Polyester polyol based PU´s are derived from dicarboxylic acids, mostly adipic acid, and generally have the following advantages over polyether polyol based PU´s
- Greater oil and solvent resistance
- Greater oxygen and UV resistance
- Improved abrasion resistance
- Increased high temperature performance
- Improved tear resistance
- Higher tensile strength
Typical applications are outlined in the charts below:
- Tabletting Applications
An excipient is any inert or inactive substance that is used in tablets as a filler, binder, lubricant or disintegrant. Adi-pure®, adipic acid from INVISTA, is ideally suited for use as an excipient in tablet manufacture as it is able to play a number of roles in the tablet.
- Paper Additives
Adipic acid is used extensively in the manufacture of polyamide-epichlorohydrin wet-strength resins. Similar products can also be manufactured by the use of dimethyl adipate or other adipate esters.
- Unsaturated Polyester Resins
Unsaturated polyester resins are the third-largest class of thermoset molding resins. The polyesters are low molecular weight viscous liquids dissolved in vinyl monomers like styrene to facilitate molding or shaping of the resin into a desired form before curing to rigid solids. Typical applications are in fiberglass-reinforced shower stalls, boat hulls, truck caps and airfoils, construction panels, and autobody parts and trim. Mineral-filled UPRs are used in synthetic marble countertops and autobody putty. Unfilled UPRs are used in gel coats and maintenance coatings. Adi-pure® adipic acid improves tensile and flexural strength in these resins and, at high levels, can give soft, pliable products for specialty applications. 1 Alkyd resins, a common type of unsaturated polyester resin, utilize adipic acid where low viscosity and high flexibility are valued in plasticizer applications.
UPR resins are mainly aromatic polyesters. Flexibility of UPR is increased by replacing a portion of aromatic acid with Adi-pure® adipic acid. A cure site monomer, like maleic anhydride, is incorporated to provide unsaturation within the polymer backbone. Crosslinking is by free radical addition polymerization of styrene monomer/diluent.
- Synthetic Lubricants
Because synthetic lubricants are composed of structurally uniform molecules with well-defined properties they offer several advantages over conventional oil and greases:
- Excellent viscosity and temperature properties
- Good high temperature oxidation and thermal stability reduce deposits
- Good low temperature characteristics improve cold flow properties
- Low volatility reduces consumption
These advantages all help to increase operational efficiency, reduce maintance costs and energy consumption.
- Chemical Intermediates
Adiponitrile Synthesis
Adiponitrile, the key intermediate for the manufacture of 1,6-hexanediamine, can be produced by catalytic dehydration of the ammonium salt of adipic acid at approximately 350°C(662°F). Yields of adiponitrile typically exceed 90%.
Cyclopentanone Synthesis
Cyclopentanone, an intermediate useful in the synthesis of flavor and fragrance chemicals, glutaric acid, and biologically active compounds, is readily produced by the calcium- or barium-catalyzed decarboxylation/dehydration of adipic acid at temperatures greater than 230°C(446°F).
1,6-Hexanediol Synthesis
Hydrogenation of dimethyl adipate over a promoted copper chromite catalyst at 200°C(392°F) and 100 kPa (750 mmHg) produces 1,6-hexanediol, a valuable chemical intermediate used in polyester polyol, urethane, and alkyd resin manufacture. Other catalysts include nickel and promoted cobalt systems.
Dimethyl Sebacate Synthesis
The Kolbe electrolysis of monomethyl adipate salts leads to the formation of dimethyl sebacate from which sebacic acid can be easily prepared. Sebacic acid is typically used in the synthesis of nylon 6,10 and alkyd resins that are flexible and act as plasticizers.
- Sequestrants
Sequestrants are employed in a diverse range of products and processes – from household products to industrial applications.
To sequester means to „withdraw from use“ and in particular sequestrants are used to sequester metal ions such as iron, copper, nickel, cobalt, chromium, calcium and manganese.
Sequestrants act by forming strongly associated but soluble complexes with metal ions thereby preventing them from precipitating out or becoming involved in other reactions. These metal ions find their way into products as a result of, for example, hardness in water, directly from the soil or as a contaminant in raw materials.
Sequestrant capability
A measure of the tendency towards formation of metal complexes in aqueous solution is the stability constant, K.
Metal Ion Stability Constant, K Ca2+ 2.19 Cd2+ 2.1 Cu2+ 2.3 Ni2+ 1.6 Pb2+ 2.8 Zn2+ 1.8 Table 1. Log of Stability constants with adipic acid at 25ºC.
Table 1 demonstrates that Adi-pure® is able to form stable metal complexes with many different metal ions.
- Solder Fluxes
Solder flux and flux-cored solders are used to improve the quality of electrical connections by cleaning connector surfaces and promoting metal-to-metal bonding. Electronics connec- tions particularly require fluxes that leave minimal residues and are readily cleaned without oxidation of the connections. Where it is desirable to avoid cleaning the connections after soldering, a "weak acid" dilute adipic acid alcohol solution is preferred. Adipic acid is used in a number of flux and flux-cored solder formulations to activate the flux, promote metal cleaning, and minimize residual impurities and corrosion. The very low trace impurity levels of Adi-pure® highpurity adipic acid make it highly desirable for highquality electronics applications.
- Leather Tanning
The primary function of a tanning agent is to stabilize collagen fibers in the fresh hide so they are not readily biodegradable. Adipic acid can be used to improve leather quality and to increase the utilization of chromium salts in tanning liquors, thereby minimizing chromium losses to effluent streams.
- Flue Gas Desulfurization Scrubbers
Adipic acid and mixed dibasic acids that contain adipic acid are routinely used to enhance the performance of wet limestone scrubbers for flue gas desulfurization. Environmental re- gulations limiting sulfur dioxide emissions are expected to increase the demand for such scrubber systems. Adipic acid acts as a buffer, improves the scrubber efficiency by increa- sing the dissolution rate of limestone into the liquid slurry fed to the scrubber, and impro- ves the mass transfer of sulfur dioxide from the gas phase to the scrubber liquid. Adipic acid is preferred in forced-air oxidation limestone scrubber operations.
- Cleaning Applications
Adipic acid and its derivatives can be used in a variety of cleaning applications, ranging from laundry detergents and bleaches to dishwashing detergents and hard-surface cleaners. Laundry detergents that contain adipic acid form watersoluble salts or chelate complexes and prevent redeposition of calcium, magnesium, and other metal salts onto fabrics. Amidoperoxyadipic acids are used for laundry bleaches that replace chlorine-based bleaches. The chelating properties of adipic acid are also valuable in formulating dishwashing detergents and hard-surface cleaners for use in bathrooms and kitchens where solubilization of calcium- and magnesium-based soap scum is particularly important.
Properties
- Physical Properties
- Typical Properties
- Thermal Properties
- Solution Properties
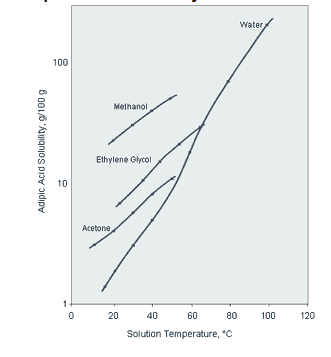
Property Typical Value Solubility see Figure below the Table pH of Aqueous
Solutions at
25°C (77°F)Concentration pH 0.1% 3.2 0.2% 3.1 0.4% 3 0.6% 2.9 1.2% 2.8 2.5% 2.7 Density of Saturated
Aqueous SolutionsTemperature Density 20°C (68°F) 1.0045 g/cc 40°C (104°F) 1.0050 g/cc 60°C (140°F) 1.0125 g/cc Ionization Constant
( K) in Water at 25°C
(77°F)Step 1 3.71 x 10–5 (p K1 = 4.43) Step 2 3.87 x 10–6 (p K2 = 5.41) Adipic Acid Solubility
- Other Typical Physical Properties
Property Typical Value Flash Point, Cleveland
Open Cup210°C (410°F) Flash Point, Tag
Closed Cup196°C (385°F) Vapor Pressure, Molten Temperature Vapor Pressure 159.5°C (319.1°F) 0.13 kPa (1 mmHg) 205.5°C (401.9°F) 1.33 kPa (10 mmHg) 240.5°C (464.9°F) 5.33 kPa (40 mmHg) 265.0°C (509.0°F) 13.33 kPa (100 mmHg) 312.5°C (594.5°F) 53.32 kPa (400 mmHg) 337.5°C (639.5°F) (with decomposition) 101.31 kPa (760 mmHg) Viscosity, Molten Temperature Viscosity 160°C (320°F) 4.54 cP 193°C (379.4°F) 2.64 cP
| Value | Units | Test Method / Conditions | |
| Molecular Weight | 146.14 | — | — |
| Boiling Point (760 mm) with Decomposition | 337.5 (639.5) | °C (°F) | — |
| Melting Point | 151.5 - 153 (304.7 - 307.4) | °C (°F) | — |
| Density (Solid) | 1.360 | g/cc | — |
| Density (Liquid at 163°C (325.4°F)) | 1.093 | g/cc | — |
| Bulk Density (Loose) | 640 - 720 (40 - 45) | kg/m³(lb/ft³) | — |
| Bulk Density (Packed) | 800 - 930 (50 - 58) | kg/m³(lb/ft³) | — |
| Hygroscopicity (at 27°C (80.6°F) and 85% RH) | Nonhygroscopic | — | — |
| Crystal Form | Monoclinic needles | — | — |
| Value | Units | Test Method / Conditions | |
| Adipic Acid Content | min. 99.7 | wt% | — |
| Water Content | max. 0.2 | wt% | — |
| Ash Content | max. 2 | ppm | — |
| Iron Content | max. 0.5 | ppm | — |
| Methanol Solution Color | max. 4 | APHA | — |
| Total Nitrogen Content | max. 15 (max. 1.5) | mpm (ppm) | — |
| Value | Units | Test Method / Conditions | |
| Specific Heat (Solid) | 1.59 (0.38) | kJ/kg·°K (cal/g·°C) | — |
| Specific Heat (Liquid) | 2.26 (0.54) | kJ/kg·°K (cal/g·°C) | — |
| Heat of Formation | -6503 (-1625.8) | kJ/kg (cal/g) | — |
| Heat of Fusion | 238.5 (57) | kJ/kg (cal/g) | — |
| Heat of Vaporization | 560.3 (133.9) | kJ/kg (cal/g) | — |
| Heat of Sublimation (at 25°C (77°F)) | 881.8 (210.8) | kJ/kg (cal/g) | — |
| Heat of Combustion | 19,149 (4576.8) | kJ/kg (cal/g) | — |
| Heat of Solution (Water 10–20°C (50–68°F)) | -213.6 (-51.1) | kJ/kg (cal/g) | — |
| Heat of Solution (Water at 90–100°C (194–212°F)) | -240.5 (-57.5) | kJ/kg (cal/g) | — |
| Heat of Crystallization from Water | 265.3 (63.4) | kJ/kg (cal/g) | — |
Regulatory & Compliance
- Certifications & Compliance
Technical Details & Test Data
- Plasticizers
A plasticizer is a substance which when added to a material, usually a plastic, makes it flexible, resilient and easier to handle. Modern plasticizers are man-made organic chemicals, the majority of which are diesters produced from high molecular weight alcohols such as adipic and phthalic acid.
Typical adipate plasticizers are:
- Dioctyladipate (DOA)
- Diisodecyladipate
- Adipate based polymeric plasticizer
Safety & Health
- Epoxy Curing Agents
Carboxy-terminated polyesters are often used as curing agents for glycidyl ether resins
and for epoxidized olefin resins. The carboxy-terminated polyester prepared from adipic acid, diethylene glycol, and glycerol is used as a curing agent for the diglycidyl ether of bisphenol A.Adipic acid and similar saturated dibasic acids are used at a mole ratio of less than 1:1 with diepoxy resins to promote etherification or are used in substantial excess with a suitable catalyst to accomplish resin cross-linking by condensation esterification.
The so-called fatty polyamides are often used for epoxy curing and can be prepared, for example, from adipic acid, ethanolamine, and ethylene glycol. Fatty polyamides impart flexibility and improved water resistance to epoxy coatings, particularly the glycidyl ether resins. Construction applications, general-purpose adhesives, and can seam bonding are typical areas of use.
Packaging & Availability
- Packaging Information
Adi-pure® can be packaged for shipment in a variety of containers, depending on customer needs. Pack sizes range from small bags to big bags. Bulk shipments - for example railcars and 27 ton Bag-in-Box - can also be supplied.

