Knowde Enhanced TDS
Identification & Functionality
- Chemical Family
- Chemical Name
- Technologies
Features & Benefits
- Labeling Claims
- Base Chemicals Features
- Product Advantages
Unlike other alumina manufacturing processes which use less pure bauxite derivatives as a starting material, Sasol has pioneered a process based on aluminium alkoxide which produces synthetic alumina hydrates of high purity. Examples of some trace impurities are shown in table 1. Sasol produces aluminium oxides with a wide range of possible particle size distributions (figure 1). The range of possible average particle sizes stretches from 8 micrometers to 2.5 millimeters. Other pore size distributions are available on request.
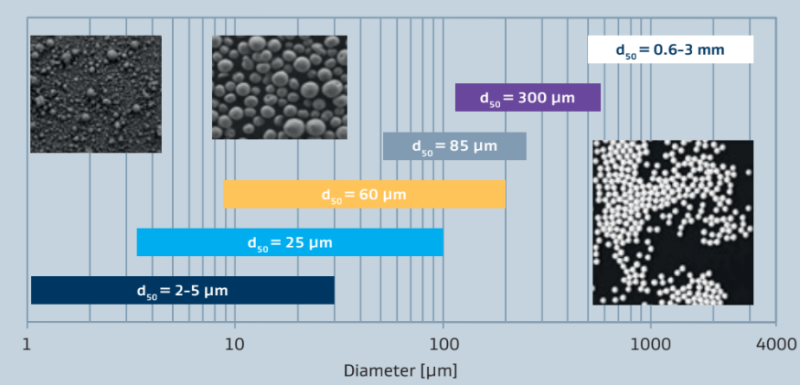
Figure 1: Areas of particle size distribution of PURALOX and CATALOX aluminas
Impurity ppm (typical) Na₂O <20 Fe₂O₃ 50-100 SiO₂ 50-120 Table 1: Chemical purity of PURALOX Aluminas
Applications & Uses
- Markets
- Base Chemicals End Uses
Properties
- Physico-Chemical Properties
| Value | Units | Test Method / Conditions | |
| Aluminium Oxide (Al₂O₃) | 80.0 | % | — |
| Sodium Oxide (Na₂O) | 0.002 | % | — |
| Zirconium Dioxide (ZrO₂) | 20.0 | % | — |
| Loss on Ignition (L.O.I.) | 3.0 | % | — |
| Loose Bulk Density | 600 - 800 | g/l | — |
| Particle Size (d50) | 30.0 | μm | — |
| Surface Area (BET) | 190.0 | m²/g | — |
| Pore Volume | 0.5 | ml/g | — |
| Pore Radius | 5.0 | nm | — |
Technical Details & Test Data
- Activation
The final crystalline phase of the activated alumina depends on the initial crystalline properties of the starting material as well as the activation process. Our activated aluminas are predominantly based on high purity boehmite as the starting alumina hydrate.
The phase transitions of boehmite are shown in figure 2 in the form of powder X-Ray Diffraction (XRD) patterns.

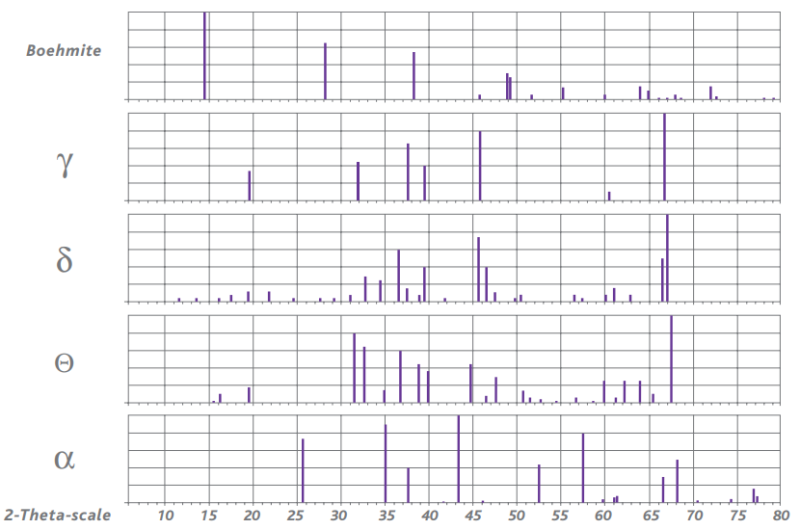
Figure 2: Powder XRD patterns of various alumina phases

Typical activation temperatures of the boehmite lie within 600–1000°C. During such temperatures the physically and chemically bound water is removed, transforming the hydrate into an oxide.
However,the use of bayerite as the starting hydrate is also available upon request. The bayerite is produced with the same high purity characteristics as the boehmite.
Properties such as the crystalline phase, surface area and porosity can be altered significantly by varying the activation process. Figure 3 shows a graphic representation of the relationship between the surface area, pore volume and crystalline phase. Depending on the physical characteristics of the initial alumina hydrate Sasol is able to prepare aluminum oxides with various pore volumes. The data shown in figure 3 are by no means the only possibilities available.
Figure 4 shows the differential scanning calorimetry (DSC) of boehmite and bayerite indicating the different Al2O3-phase conversions and the corresponding conversion temperatures.
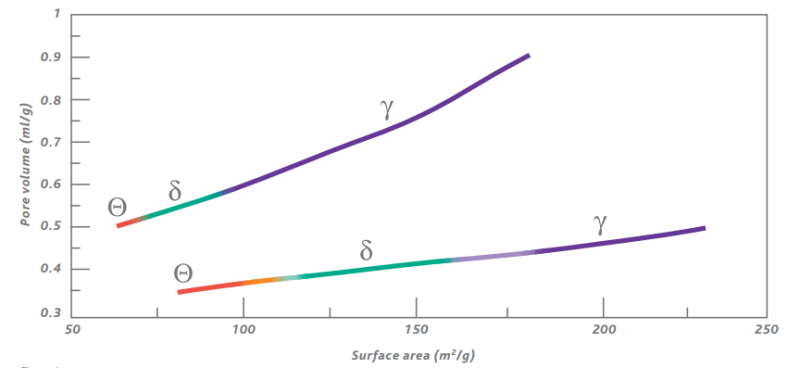
Figure 3: Example of the relationship between surface area,pore volume and crystal phase
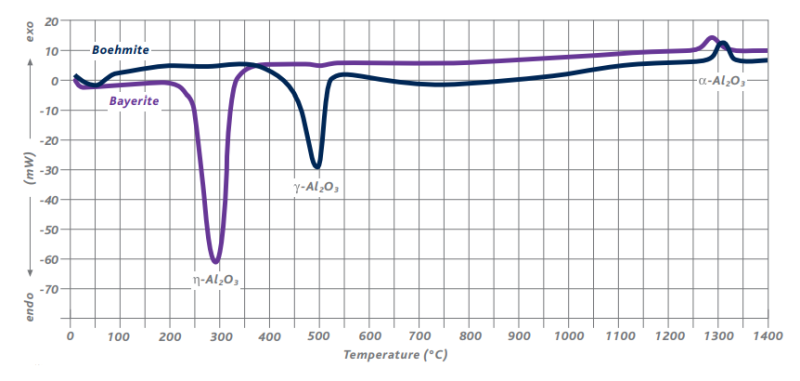
Figure 4: DSC of boehmite and bayerite aluminas
- Calcination
The final crystalline phase and physical properties of the alumina depend on the physical properties of the starting material as well as the calcination process. Our calcined aluminas are predominantly based on high-purity boehmite as the starting alumina hydrate. Typical calcination temperatures of the boehmite lie within 600-1000 °C. Applying such temperatures, the physically and chemically bound water is removed, transforming the hydrate into an oxide. High temperature calcination leading to α-Alumina can also be applied.
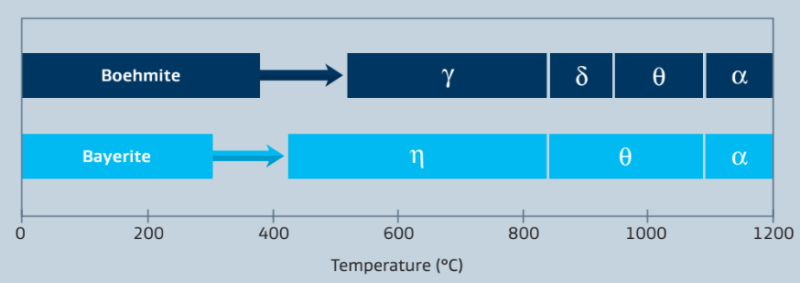
Figure 5: Sequence of phase transistions
Thy physical properties of the aluminum oxides are to a large extend adjusted at the stage of the corresponding hydrates by varying their crystallite size and shape. This way, aluminas with tailored porosities are accessible, ranging from high-density to high-porosity materials. These two border cases are illustrated in Figure 6.
Properties such as the crystalline phase, surface area and porosity can be altered signifi- cantly by varying the calcination process. Figure 7 shows a graphic representation of the relationship between the surface area, pore volume and average pore radius. Depending on the physical characteristics of the initial alumina hydrate Sasol is able to prepare aluminum oxides with various pore volumes.

Figure 6: Electron tomography of a) high-density b) high-porosity aluminas showing different crystallite arrangements.
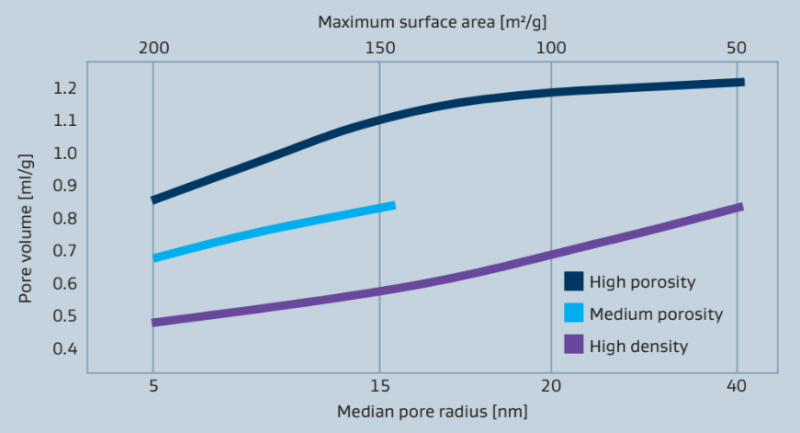
Figure 7: Alumina grades with different porosities
- Analytical Methods
- Trace element analysis - Alumina powder is quantitatively brought into solution by using acids and then analyzed by ICP, atomic emission. Additionally, X-ray fluorescence spectroscopy is used.
- Crystallite type and average crystal size - Powdered samples of the alumina are analyzed by using X-Ray Diffractometry (XRD) on either a Siemens D5000 or a Philips X’Pert diffractometer. The resulting diffractogram enables the laboratory to identify the crystal structure of the material.
- Particle size distribution - The particle size distribution of alumina may be measured by various instruments, namely, Cilas Granulometer 1064 supplied by Quantachrome, Malvern Mastersizer or Luftstrahlsieb (air sieve) supplied by Alpine.
- Surface area analysis - The surface area of the alumina is measured by using an instrument supplied by Quantachrome (Nova series) or by Micromeritics (Gemini series). The method entails low temperature adsorption of nitrogen at the BET region of the adsorption isotherm.
- Pore size distribution - The pore size of our products is measured by two different methods. Measurement may be performed by either mercury intrusion or by nitrogen desorption. Nitrogen desorption, with instruments from Quantachrome, is used to find the pores from 2 to 30 nm while the mercury intrusion is used to obtain the pores between 5 to 500 nm using an Micromeritics Autopore instrument.
- Differential scanning calorimetry (DSC) - Netzsch STA 449C Jupiter, Setaram 92 or Perkin Elmer instruments may be used with a selected heating rate to obtain the exothermic and endothermic transitions of alumina. Additional test methods are available for other physical properties upon request.
- Doped Aluminas
Sasol aluminas are also available as doped or even multi-doped versions (e.g. La, Ce, Ti, Zr). The resulting interplay of well-defined physical properties and chemical modifications is a further step towards optimized support materials for various applications like emission control or refinery catalysts. As an example, the addition of lanthanum-oxide improves the thermostability of the base alumina as it can be seen in figure 9.
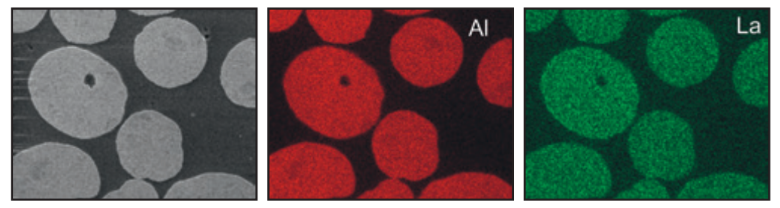
Figure 8: SEM-EDX illustration of a La-doped alumina
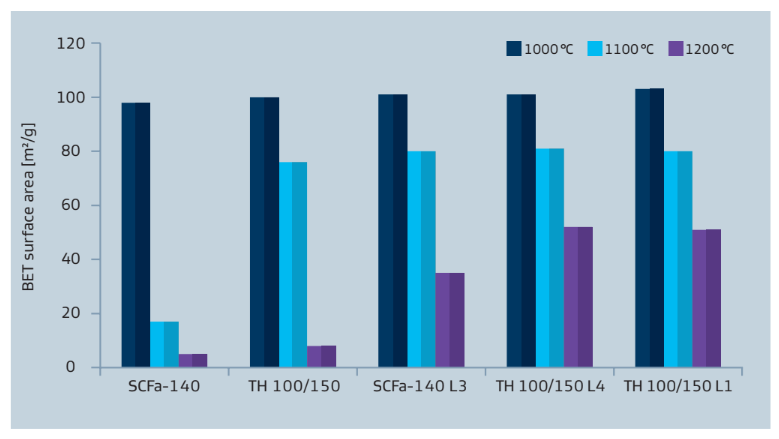
Figure 9: Thermostability of pure, La- and Ba-doped aluminas
Safety & Health
- Safety Information
- PURALOX aluminas are classified as a non-toxic, non-flammable nuisance dust. Exposure to high concentrations of dust may cause physical irritation. Repeated or prolonged exposure with skin may result in dryness and irritation.
- Handling procedures should be designed to minimize inhalation and skin exposure. Normal good housekeeping and operating procedures should ensure personnel safety.
Storage & Handling
- Storage and Transfer Conditions
Unlike other alumina manufacturing processes which use less pure bauxite derivatives as a starting material, Sasol has pioneered a process based on aluminum alkoxide which produces synthetic alumina hydrates of high purity. Examples of some trace impurities are shown in table 1. Sasol produces aluminum oxides with a wide range of possible particle size distributions (figure 1). The range of possible average particle sizes stretches from 8 micrometers to 2.5 millimeters. Other pore size distributions are available on request.

Figure 1: Areas of particle size distribution of PURALOX and CATALOX aluminas
Impurity ppm (typical) Na₂O <20 Fe₂O₃ 50-100 SiO₂ 50-120 Table 1: Chemical purity of PURALOX Aluminas

