Knowde Enhanced TDS
Identification & Functionality
- Enzyme Type
- Ingredient Name
- Ingredient Origin
- Food Ingredients Functions
- Ingredients
- Water, Glycerol, Potassium Sorbate, Fructosyltransferase, Sugar Syrup
- Technologies
Features & Benefits
- Labeling Claims
- Food Ingredients Features
Applications & Uses
- Markets
- Food & Nutrition Applications
Properties
- Physical Form
- Appearance
- Liquid
- Chemical Properties
- Typical Properties
- Microbiological Values
| Value | Units | Test Method / Conditions | |
| Arsenic Content | max. 3 | mg/kg | AAS |
| Cadmium Content | max. 0.5 | mg/kg | AAS |
| Lead Content | max. 5 | mg/kg | AAS |
| Mercury Content | max. 0.5 | mg/kg | AAS |
| Value | Units | Test Method / Conditions | |
| Activity | min. 5000 | u/g | — |
| Value | Units | Test Method / Conditions | |
| Escherichia coli | max. 10 | cfu/g | §64 LFGB |
| Salmonella | Negative | cfu/25g | §64 LFGB |
| Total Plate Count | max. 50000 | cfu/g | §64 LFGB |
Regulatory & Compliance
- Regulatory Information
Allergen Information: no substances or products causing allergies or intolerances according to Regulation 1169/2011/EG, Annex II
Health Information: The products are suitable for human consumption if used accordingly to recommendations and officially allowed to be sold within Germany and the EU. The product is food grade and conforms with the current european food law.\
GMO: The components of this product do not contain and are not derived from genetically modified organisms. The product is not subject to GMO-labeling and traceability according to Regulations (EC) No. 1829/2003 and 1830/2003.
Product suitability:
Yes No Vegetarian X Vegan X BSE/TSE Information: The mentioned product does not represent a BSE (Bovine Spongiform Encephalopathy) and/or TSE (Transmissible Spongiform Encephalopathy) risk.
Irradiation: Neither the product nor its ingredients have been treated with ionizing radiation according to Directive 1999/2/EC and 1999/3/EC.
Nanoparticles: The product does not contain ingredients in the form of engineered nanomaterials as defined in Regulation (EU) 2015/2283.
Contamination/Residues: The product complies with: Regulation (EC) No 396/2005 and its amendments regarding maximum levels for pesticide residues in foodstuffs. Regulation (EC) No 1881/2006 and its amendments regarding especially maximum levels for heavy metals, mycotoxins, dioxins and PCB in foodstuffs.
Conformity of the Packaging: The packaging material complies with the provisions of Regulation (EC) No 1935/2004 and Regulation (EU) No 10/2011.
Technical Details & Test Data
- Prebiotic fructooligosaccharides (FOS)
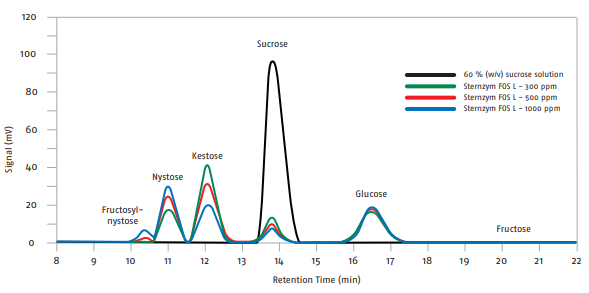
Sternzym FOS L catalyzes the hydrolysis of sucrose and subsequently the transfer of fructosyl-groups to a sucrose molecule or to an existing FOS. Depending on the enzyme dosage and reaction time, mainly Kestose (GF2), Nystose (GF3) and Fructosylnystose (GF4) are formed (see Fig. 1). A total conversion rate of sucrose to fructose of 50 to 60 % can be reached.
Fig. 1 HPLC analysis of the saccharide composition after treatment with Sternzym FOS L (pH 5.5, 55 - 60 °C, 16 h).
Application of Sternzym FOS L

1. A 60% (w/v) sucrose solutions is used as a starting material for the production of prebiotic FOS.
2. After the complete dissolution of sucrose, the pH should be adjusted to a value of 5.0 to 5.5. The temperature should be set to 55 to 60°C.
3. Prepare a 10% enzyme solution.
4. To this sucrose solution, 3 ml of the 10% Sternzym FOS L solution should be added. This corresponds to a level of 300 ppm per syrup.
5. Incubate for 16 to 24h.
Packaging & Availability
- Packaging Type
- Packaging
Can, 25 kg net weight, futher packaging units on request
Storage & Handling
- Storage & Shelf Life
Min. 24 months if stored cool (< 10°C) and dry in closed original packing

