Knowde Enhanced TDS
Identification & Functionality
- Chemical Name
- Pharma & Nutraceuticals Functions
- CAS No.
- 9063-38-1
- EC No.
- 618-597-7
- Technologies
Features & Benefits
- Benefits of Superdisintegrants
Our superdisintegrant offerings meet the requirements necessary to achieve fast and robust disintegration with minimal amount of disintegrating agent. These properties facilitate the use of any ingredient you need in order to optimize other parameters of your formulation (compactability or drug solubility for example).
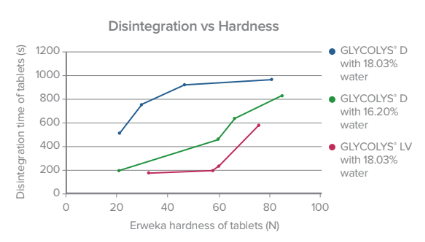
GLYCOLYS®, one of our sodium starch glycolate (SSG)-based products, offers solutions to specific environments and conditions susceptible to affect disintegrant performance as well as ensuring stability for drugs, like GLYCOLYS® Low pH ensures stability to acidic molecules.
Applications & Uses
- Markets
- Manufacturing Technology
- Use Level
- 2 - 8%
Properties
- Appearance
- White fine powder
- Insoluble in
- Methylene Chloride
- Typical Properties
- Microbiological Values
| Value | Units | Test Method / Conditions | |
| Combined Sodium Content | 2.8 - 4.2 | % | Ph.Eur.-USP-JP |
| Ethanol Content | max. 6 | % | Ph.Eur.-USP |
| Heavy Metals Content | max. 20 | mg/kg | JP |
| Iron Content | max. 20 | mg/kg | Ph.Eur.-USP-JP |
| Loss on Drying | max. 10 | % | Ph.Eur.-USP-JP |
| Particle Size (Retained on 200 mesh. 75 microns) | max. 10.0 | % | — |
| pH | max. 10.0 | — | — |
| Sodium Chloride Content | max. 7.0 | % | Ph.Eur.-USP-JP |
| Sodium Glycolate | max. 2.0 | % | Ph.Eur.-USP-JP |
| Water Content | max. 10 | % | — |
| Value | Units | Test Method / Conditions | |
| Escherichia coli | Not Detected | per gram | — |
| Pseudomonas aeruginosa | Not detected | per gram | — |
| Salmonella | Not Detected | per 10g | — |
| Staphylococcus aureus | Not detected | per gram | — |
| Total Aerobic Microbial Count | max. 100 | CFU/g | — |
| Total Yeasts and Moulds Count | max. 10 | CFU/g | — |
Regulatory & Compliance
- Certifications & Compliance
- Chemical Inventories
- Grade
- Conformity
Complies with the current USP/NF,EP and JP monographs requirements
Technical Details & Test Data
- Innovation Hub
- Innovation Hub by RoquetteLooking for technical support or formulation inspiration? Visit Roquette’s Innovation Hub.
Packaging & Availability
- Packaging Information
- ROQ Product Code: 731801
- Article (SKU) Code: 731801551
- Package Size & Type: 30 kg lined cardboard box
Storage & Handling
- Shelf Life
- 5 Years
- Storage and Handling
Storage in well-closed containers preferably protected from wide variations in temperature and humidity, which may cause caking

