Knowde Enhanced TDS
Identification & Functionality
- Cosmetic Ingredients Functions
- Technologies
- Product Families
- Origin
- DermalRx® CKW is a proprietary extract of the rhizomes of two related species in the Curcuma genus: Curcuma kwangsiensis and Curcuma wenyujin.
Features & Benefits
- Benefit Claims
- Labeling Claims
- Benefits & Claims
- Plant-derived active produced from two exotic Zingiberaceae (ginger) species in the Curcuma genus
- Inhibits types I and II 5α-reductase activities
- Inhibitory efficacy against 5α-reductase activities is comparable to benchmark (finasteride)
- Provides antioxidant activity against environmental stress
- Well-documented minimization of sebum production in vivo among male & female populations
- Clinically proven visible decrease of facial sheen
Applications & Uses
- Markets
- Color Cosmetic Applications
- Skin Care Applications
- Use Level
- 0.5 - 1%
- Applications
- Sebum control products for oily-prone skin
- Formulas to address blemishes and dry skin
- Pore-minimizing and mattifying anti-aging formulations
- BB creams, CC creams, and color formulations (primers, foundations, powders…)
- Skincare regimens for men
- Sebum-regulating hair care products
- Compatibility and Formulation
- DermalRx® CKW is soluble in water and hydroglycolic solutions. It is compatible with conventional ingredients used in skincare products. Add to product at temperatures below 50°C.
Properties
- Physical Form
Regulatory & Compliance
- Certifications & Compliance
- Regulatory Information
- This product has not been tested on animals, and our company adheres to the written provisions of such animal rights organizations as Leaping Bunny and Cruelty/Free International.
- This product contains no animal products. It is manufactured using only plant based or synthetically derived (non-animal origin) materials and is thereby declared a Vegan Product.
- All materials used to produce this product are procured using sustainable sourcing practices.
- This product is manufactured in accordance with the current Good Manufacturing Practices (cGMP) requirements established by the U.S. Food and Drug Administration (FDA) for cosmetics. Furthermore, Biocogent, LLC, and this production facility(ies), are registered as cosmetic manufactures with the FDA.
- REACH compliance: All of the ingredients (substances) used in the manufacture of this product (mixture)are either registered under REACH, or qualify for an Article 2 (low volume of less than 1 metric ton), Annex IV or Annex V exemption, making our product REACH compliant.
- Proposition 65 compliance: Based upon Biocogent LLC’s knowledge of our business operations and chemical formulations, no chemicals listed by the State of California Environmental Protection Agency are used in our products, or as a processing aid for any product manufactured in our facility.
Technical Details & Test Data
- Test Data
Reduction of Surface Shine
DermalRx® CKW visibly reduces the appearance of shine and pore size after 4 weeks of applying 1% DermalRx® CKW.
Inhibition of Type I 5α-reductase
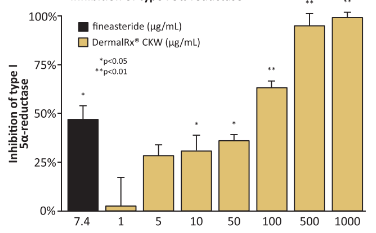
DermalRx® CKW dose-dependently inhibits 5α-reductase to help control sebum production in the skin.
Safety & Health
- Safety
- Human Repeat Insult Patch Testing (HRIPT) under semi-occlusive conditions indicated that DermalRx® CKW did not perform as a primary skin irritant or as a primary skin sensitizer at recommended use levels.
Storage & Handling
- Shelf Life
- 24 Months
- Storage Condition
- DermalRx® CKW should be stored at room temperature in original packaging away from heat and light. Protect from freezing.

