Knowde Enhanced TDS
Identification & Functionality
- Country of Origin
- Ingredient Name
- Food Ingredients Functions
- Ingredients
- Waxy Maize Starch
- Technologies
- Product Families
- Product Label
Package labeling Hydroxypropyl distarch phosphatePackage labeling US Food starch modifiedIngredients US [Listed in Descending Order] 100% (ds) Modified food starch
Features & Benefits
- Labeling Claims
- Food Ingredients Features
- Functionality
- Exceptional Cold Storage And Freeze Thaw Stability
- Superior Performance In Continuous, High Shear Systems
- Resistant To High Heat Conditions
- Can Be Used In High And Low Ph Food Systems
- Smooth, Short Texture
- Exceptional Paste Clarity
- Bland Flavor
- Extremely Bland Flavor
Applications & Uses
- Markets
- Applications
- Food & Nutrition Applications
- Application
The product is a viscosifier and stabilizer designed for food systems requiring moderate to severe processing conditions, such as :
- Pie Fillings
- Bakery Cream
- Uht Dairy Desserts (Indirect Heating)
- Frozen Desserts
- Canned Soups And Sauces
- Frozen Soups And Sauces
- Salad Dressings And Creams
Properties
- Appearance
- White to cream coloured powder
- Typical Properties
- Nutritional Information (per 100 gm)
- Note for Nutritional Data
- The list comprises relevant nutritional components only.
- Values are calculated based on the average of product specifications. In those cases where only a minimum or a maximum value is specified, these values were taken respectively.
- The EU column lists the nutritional values in accordance with Regulation (EU) n° 1169/2011 on food information to consumers.
- The US column lists the nutritional values in accordance with Code of Federal Regulations (CFR 21).
- The Japan column lists the nutritional values in accordance with the Japanese Legislation.
- The energy values may differ per region because of different calculations.
| Value | Units | Test Method / Conditions | |
| Ash Content | max. 0.5 | % | ASTM D 482 |
| Value | Units | Test Method / Conditions | |
| Energy | 353.1 | kcal/100g | — |
| Energy | 1500 | kJ/100g | — |
| Protein Content | 0.3 | g/100g | — |
| Carbohydrates | 88 | g/100g | — |
| Sugar Content | 0 | g/100g | — |
| Starch | 88 | g/100g | — |
| Fat Content | 0.1 | g/100g | — |
| Saturated Fat | 0 | g/100g | — |
| Trans Fat | 0 | g/100g | — |
| Cholesterol | 0 | mg/100g | — |
| Sodium Content | 10 | mg/100g | — |
| Salt | 25 | mg/100g | — |
| Calcium Content | 5 | mg/100g | — |
| Iron Content | 0.2 | mg/100g | — |
| Vitamin C | 0 | mg/100g | — |
| Water Content | 12 | g/100g | — |
| Added Sugars | 0 | g/100g | — |
| Dietary Fiber | 0 | g/100g | — |
| Potassium Content | 0 | mg/100g | — |
| Vitamin D | 0 | mcg/100g | — |
Regulatory & Compliance
- Certifications & Compliance
- Chemical Inventories
- Dietary Information
Suitable for Certified Halal Yes Yes Kosher Yes Yes Lacto-vegetarian Yes No Ovo-vegan Yes No Vegan Yes No Vegetarian Yes No - GMO Statement
This Cargill ingredient complies with the EU GMO requirements as principally laid down under EC Regulation No 1829/2003 on 'genetically modified food and feed' and EC Regulation 1830/2003 on 'the traceability and labeling of food and feed products produced from GMO's. Therefore, there is no need to label this product under either 1829/2003 or 1830/2003. For its operations in Europe, Cargill complies with the EU GMO requirements as principally laid down under EC Regulation No 1829/2003 on 'genetically modified food and feed' and EC Regulation No 1830/2003 on 'the traceability and labeling of food and feed products produced from GMO's'. By ensuring the supply of conventional ingredients in the EU, Cargill thus ensures that there is no need to label its products under either 1829/2003 or 1830/2003.
- Legal Requirements
- REGULATION (EC) No 852/2004 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 29 April 2004 on the hygiene of foodstuffs (as amended)
- REGULATION (EC) NO 396/2005 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 23 February 2005 on maximum residue levels of pesticides in or on food and feed of plant and animal origin (as amended)
- Status pursuant to USDA 7 CFR Part 66 National Bioengineered Food Disclosure Standard (“BE” status): The product has been sourced from a non-bioengineered source and the inadvertent presence of BE is less than 0.9%.
- Current JECFA Monograph
- REGULATION (EC) No 1935/2004 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 27 October 2004 on materials and articles intended to come into contact with food (as amended)
- REGULATION (EC) No 178/2002 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 28 January 2002 laying down the general principles and requirements of food law, establishing the European Food Safety Authority and laying down procedures in matters of food safety (as amended)
- This product is in compliance with : FDA 21 CFR § 172.892, Food starch-modified
- COMMISSION REGULATION (EC) No 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs (as amended)
- Current Food Chemical Codex
- Where manufactured in the EU this product is in compliance with Regulation (EC) 1333/2008 and subsequent amendments - on food additives.
- Regulation 231/2012 laying down specifications for food additives listed in Annexes II and III to Regulation (EC) No. 1333/2008.
Technical Details & Test Data
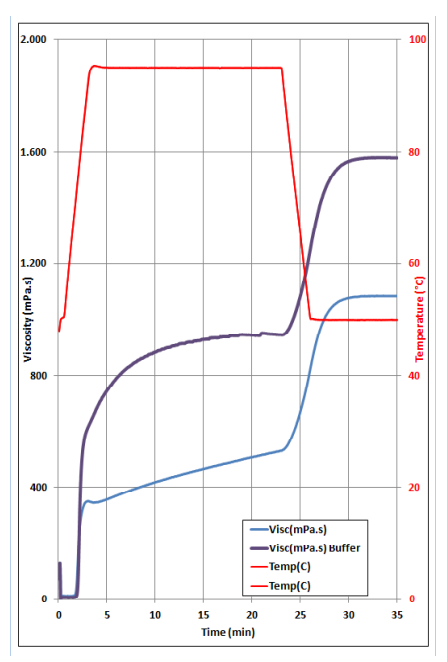
- Test Data
Packaging & Availability
- Standard Packaging
- Bulk
- Paper Bags
- Polyethylene bags
- Big Bags
Storage & Handling
- Shelf Life
- 24 Months
- Recommended Storage Conditions
- Store inside, under dry conditions
- Minimum remaining shelf life after delivery (months) : 6
- The specified shelf life can only be guaranteed for this product if the above mentioned recommended storage conditions are respected. For products delivered in bulk, there should be a clean storage (and circulation) system, protected from any potential contamination.

